Cohort studies vs case-control studies: design, bias and interpretation
Ah, the classic epidemiology showdown! Understanding the difference between cohort and case-control studies is absolutely high-yield for your USMLE prep. It really comes down to where you start and which direction you're looking.
Let me pull up some key lessons that break these designs down in detail.
📚 Study Design Lessons
To make this really clear, I've put together a comparison table that breaks down the design, the measures we use, and the common pitfalls like bias.
| Feature | Case-Control Study | Cohort Study |
|---|---|---|
| Starting Point | Outcome (Sick vs. Not Sick) | Exposure (Exposed vs. Not Exposed) |
| Direction | Retrospective (Looking back) | Prospective or Retrospective (Looking forward) |
| Measure | Odds Ratio (OR) | Relative Risk (RR) |
| Main Bias | Recall Bias, Selection Bias | Loss to Follow-up, Selection Bias |
| Best For | Rare diseases, long latency | Rare exposures, multiple outcomes |
| Interpretation | "What are the odds that the sick people were exposed?" | "What is the risk of getting sick if you're exposed?" |
Visualizing the timeline is usually the best way to keep these straight. Let me find a diagram for you that shows how these studies move through time.
Here's a great visual representation of how these studies move through time.
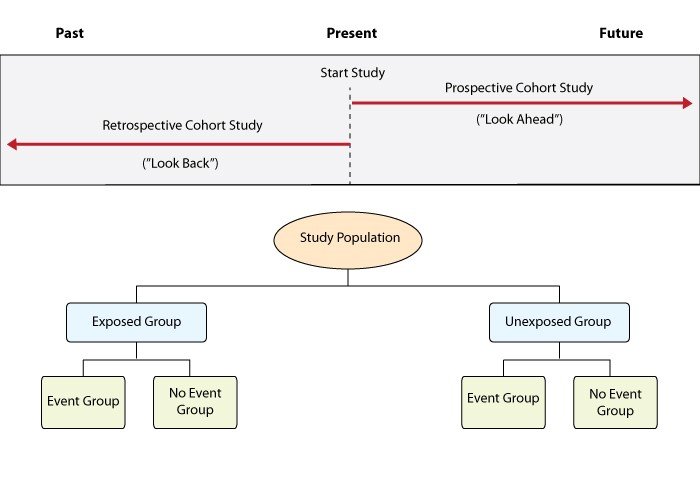
This diagram illustrates the key principles of cohort study designs, showing how we start with exposure and look forward (or backward) to see the outcome.
Key Differences in Bias & Interpretation
A quick pro-tip for your exams: if the question starts with people who are already sick, it's almost always a Case-Control study. If it starts with a group and follows them to see if they get sick, you're looking at a Cohort study.
1. Bias Breakdown:
- Recall Bias (Case-Control): Patients with a disease (cases) are more likely to remember past exposures than healthy controls. "I have lung cancer, so I remember every cigarette I ever smoked."
- Loss to Follow-up (Cohort): Since these studies can take years, people drop out, move away, or die of other causes, which can skew your results.
2. Interpretation (The Math):
- ==Odds Ratio (OR) is for Case-Control==: It tells you the odds that a person with the disease was exposed compared to someone without it.
- ==Relative Risk (RR) is for Cohort==: It tells you how much more likely an exposed person is to get the disease compared to an unexposed person.
Want to lock this in? I can pull up some flashcards or a quick quiz to test your knowledge on these concepts!
Setting up chat...