Acid-base balance: Henderson-Hasselbalch equation and clinical disorders
Acid-base balance is one of those topics that can feel like a math class in the middle of a hospital ward, but it's actually the secret to understanding how our body keeps everything in perfect harmony. At the heart of it all is the Henderson-Hasselbalch equation, which helps us calculate the pH of a buffer solution and understand how the lungs and kidneys work together.
Let me find a good visual to help us visualize this balance before we dive into the clinical disorders.
That diagram perfectly captures the "seesaw" nature of pH. The Henderson-Hasselbalch equation is essentially the mathematical version of that seesaw:
$$pH = pK_a + \log \frac{[\text{Base}]}{[\text{Acid}]}$$
In the human body, we use the bicarbonate buffer system, so it looks like this:
$$pH = 6.1 + \log \frac{[HCO_3^-]}{0.03 \times PCO_2}$$
- $pH$: The target is usually
7.4(range 7.35–7.45). - $HCO_3^-$ (Bicarbonate): Regulated by the kidneys (the metabolic component).
- $PCO_2$ (Partial pressure of $CO_2$): Regulated by the lungs (the respiratory component).
I'll show you that diagram now so you can keep the visual in mind as we talk about what happens when things go wrong.
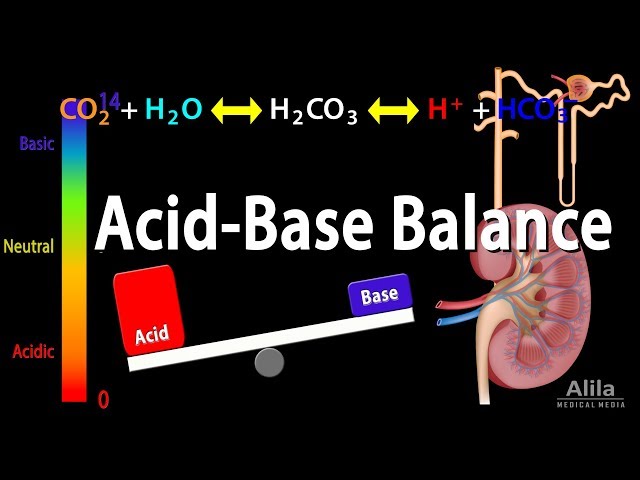
When the balance shifts, we get clinical disorders. The easiest way to remember them is to look at which part of the equation is changing first (the "primary" change) and how the body tries to fix it (the "compensation").
Here's a quick breakdown of the four main disorders:
| Disorder | Primary Change | Compensatory Response | Common Causes |
|---|---|---|---|
| Metabolic Acidosis | $\downarrow HCO_3^-$ | $\downarrow PCO_2$ (Hyperventilation) | DKA, Diarrhea, Renal failure |
| Metabolic Alkalosis | $\uparrow HCO_3^-$ | $\uparrow PCO_2$ (Hypoventilation) | Vomiting, Diuretics, Antacids |
| Respiratory Acidosis | $\uparrow PCO_2$ | $\uparrow HCO_3^-$ (Renal retention) | COPD, Opioid overdose, Obesity |
| Respiratory Alkalosis | $\downarrow PCO_2$ | $\downarrow HCO_3^-$ (Renal excretion) | Anxiety, High altitude, Salicylates |
High-Yield Tip: The Anion Gap In metabolic acidosis, always calculate the Anion Gap ($AG = Na^+ - (Cl^- + HCO_3^-)$).
- High AG: MUDPILES (Methanol, Uremia, DKA, Propylene glycol, Iron/INH, Lactic acidosis, Ethylene glycol, Salicylates).
- Normal AG: HARDUPS (Hyperalimentation, Acetazolamide, Renal tubular acidosis, Diarrhea, Ureteroenteric fistula, Pancreatic fistula, Spironolactone).
I'll pull up some lessons so you can dive deeper into the compensation mechanisms and ABG interpretation.
📚 Acid-Base Balance Lessons
Those lessons are fantastic for mastering the compensation rules—like Winter's Formula for metabolic acidosis ($PCO_2 = 1.5 \times [HCO_3^-] + 8 \pm 2$). If the measured $PCO_2$ is higher than the calculated value, you've got a secondary respiratory acidosis!
To really lock this in, would you like to try a few practice questions? I can pull up a quiz focused on acid-base disorders for you.
✍️ Acid-Base Balance Quiz
Tap to practice questions in the Oncourse app
That quiz should give you a good sense of how these concepts show up in exams—especially those tricky ABG interpretation questions!
If you're feeling confident with the theory, I can also set up a Clinical Rounds game where we walk through a real patient case with acid-base issues. Just let me know!
Setting up chat...