CO₂ Transport Forms - The Three‑Lane Highway
-
Bicarbonate (HCO₃⁻): ~70%
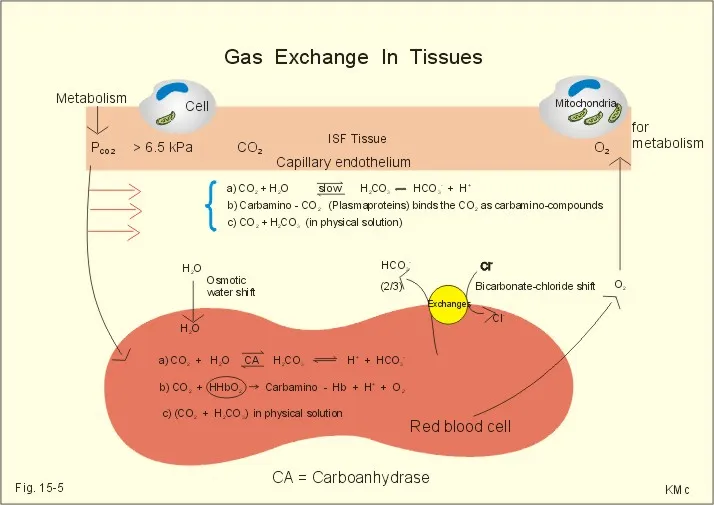
- The primary transport method. In RBCs, carbonic anhydrase catalyzes the reaction: $CO_2 + H_2O \leftrightarrow H_2CO_3 \leftrightarrow H^+ + HCO_3^-$.
- HCO₃⁻ is then transported into the plasma in exchange for chloride ions (Cl⁻), a process known as the chloride shift or Hamburger effect.
-
Carbaminohemoglobin (Hb-CO₂): ~23%
- CO₂ binds directly to the N-terminal globin chains of deoxyhemoglobin, not to the heme iron.
-
Dissolved CO₂: ~7%
- A small fraction is transported freely dissolved in plasma.
⭐ Haldane Effect: In peripheral tissues, deoxygenated hemoglobin has a higher affinity for CO₂. Conversely, in the lungs, O₂ binding to hemoglobin promotes the release of CO₂.
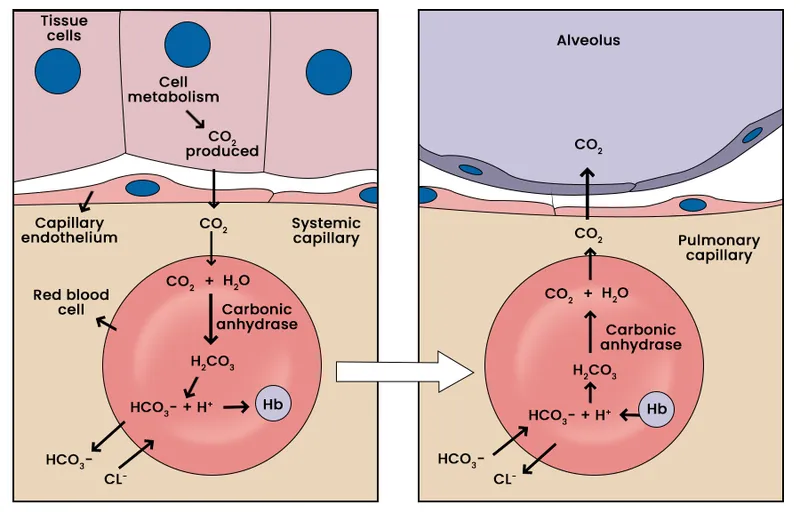
Chloride Shift - The Bicarb Swap
- In peripheral tissues with high $CO_2$, $CO_2$ diffuses into Red Blood Cells (RBCs).
- Carbonic anhydrase rapidly catalyzes the reaction: $CO_2 + H_2O \rightleftharpoons H_2CO_3$.
- Carbonic acid ($H_2CO_3$) dissociates into $H^+$ and bicarbonate ($HCO_3^-$).
- To maintain electrical neutrality, an anion exchanger (Band 3 protein) pumps $HCO_3^-$ out into the plasma in exchange for $Cl^-$ moving into the RBC.
- This 1:1 exchange is the Chloride Shift (or Hamburger phenomenon).
⭐ The Chloride Shift reverses in the lungs (a reverse chloride shift). As $CO_2$ is exhaled, plasma $HCO_3^-$ re-enters the RBC in exchange for $Cl^-$, allowing it to be converted back to $CO_2$.
Haldane Effect - Oxygen's Influence
-
Core Principle: Deoxygenated hemoglobin binds CO₂ more readily than oxygenated hemoglobin. This effect facilitates CO₂ transport from tissues to lungs.
-
Mechanism of Action:
- Tissues (↓O₂): O₂ release from Hb promotes binding of H⁺ and CO₂ (as carbaminohemoglobin), increasing blood's CO₂ capacity.
- Lungs (↑O₂): Hb oxygenation causes release of H⁺, which drives the conversion of bicarbonate to CO₂ for exhalation. $H⁺ + HCO₃⁻ → CO₂ + H₂O$
⭐ The Haldane effect accounts for about 50% of total CO₂ transport, making it a major factor in gas exchange.
CO₂ Dissociation Curve - The Other Curve
- Plots total CO₂ content of blood vs. $PCO_2$.
- Shape is nearly linear in the physiological range, unlike the O₂-Hb curve.
- Haldane Effect: Deoxygenation of Hb increases its ability to carry CO₂.
- Curve shifts up/left for deoxygenated blood (tissues).
- Curve shifts down/right for oxygenated blood (lungs).
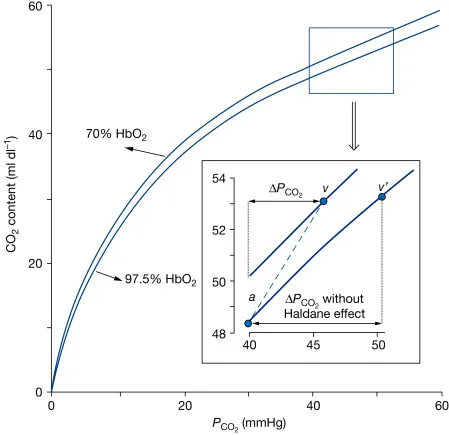
⭐ The Haldane effect has a greater quantitative impact on CO₂ transport than the Bohr effect has on O₂ transport.
High‑Yield Points - ⚡ Biggest Takeaways
- Most CO₂ is transported as bicarbonate (HCO₃⁻) (~70%), a reaction catalyzed by carbonic anhydrase within RBCs.
- The chloride shift maintains electrical neutrality by exchanging HCO₃⁻ for Cl⁻ across the RBC membrane in peripheral tissues.
- Haldane effect: Oxygenation of hemoglobin in the lungs promotes the dissociation of H⁺ and CO₂, facilitating CO₂ excretion.
- Bohr effect: In peripheral tissues, high CO₂ and H⁺ levels decrease hemoglobin's affinity for O₂, enhancing O₂ delivery.
- Carbaminohemoglobin (~23%) and dissolved CO₂ in plasma (~7%) are the other significant transport forms.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

