Genetic Testing in Pediatrics — MCQs

Phenotypic expression of a gene depending on the parent of origin is referred to as:
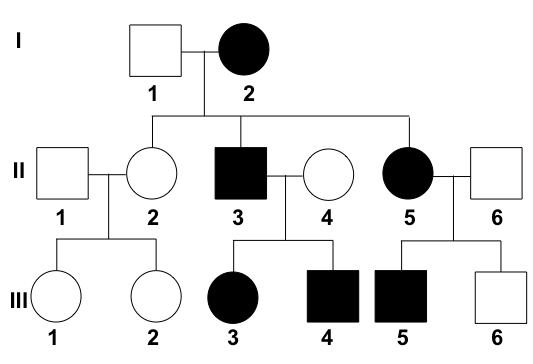
What is the interpretation of the given pedigree chart?
All of the following are tests done for Turner mosaic screening except?
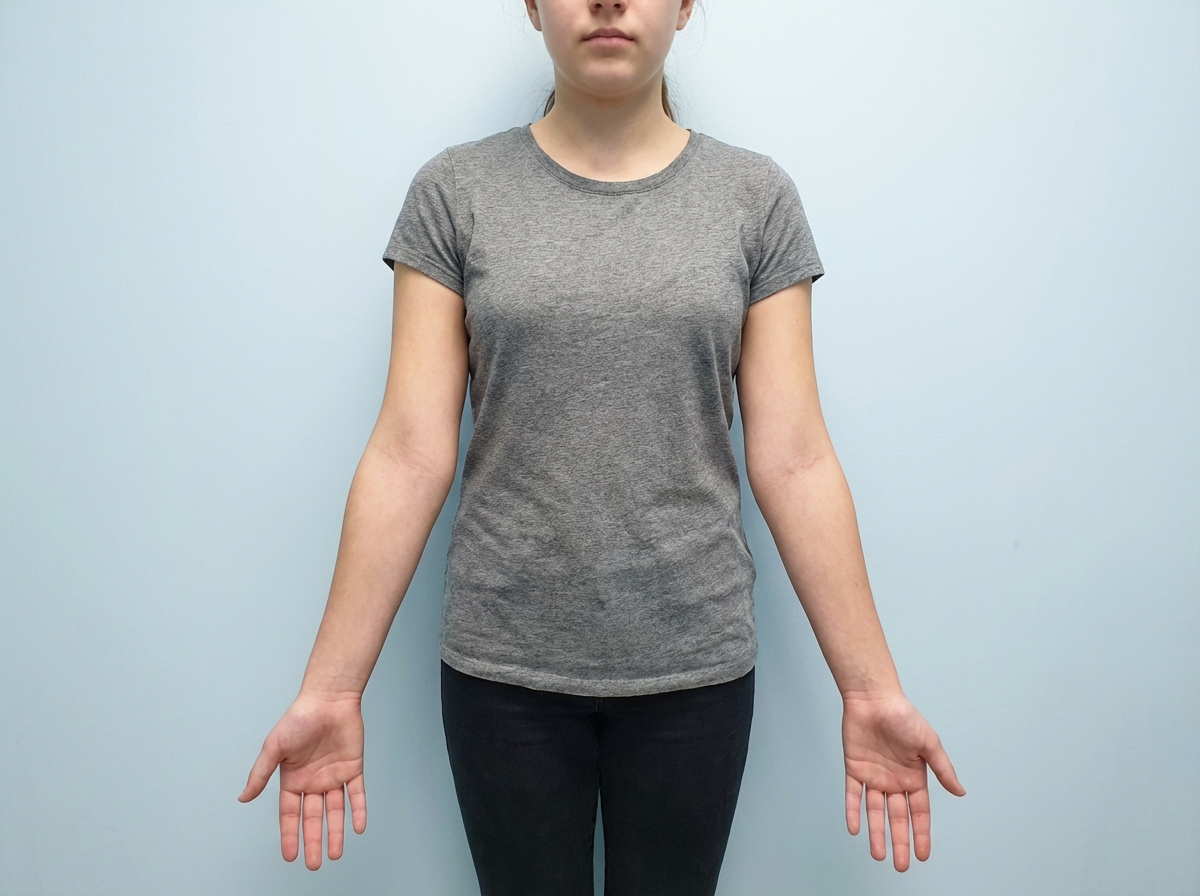
A teenage girl presents with a history of amenorrhea. Local examination is shown in the image. What karyotype analysis would you consider for further evaluation?
A baby's blood group was determined as O negative. Which of the following blood groups can the baby's mother or father not have?
34 week primigravida punjabi khatri comes with history of consanguineous marriage, with history of repeated blood transfusion to her sibling since 8 months of age. The first diagnostic test is -
A 4-year-old boy is brought to a pediatrician by his parents for a consultation after his teacher complained about his inability to focus or make friends at school. They mention that the boy does not interact well with others at home, school, or daycare. On physical examination, his vital signs are stable with normal weight, height, and head circumference for his age and sex. His general examination and neurologic examination are completely normal. A recent audiological evaluation shows normal hearing, and intellectual disability has been ruled out by a clinical psychologist. Which of the following investigations is indicated as part of his diagnostic evaluation at present?
What is the chance of a child having cystic fibrosis if both parents are carriers of the disease?
A 34-year-old gravida 2 para 1 woman at 16 weeks gestation presents for prenatal care. Her prenatal course has been uncomplicated. She takes no medications besides her prenatal vitamin which she takes every day, and she has been compliant with routine prenatal care. She has a 7-year-old daughter who is healthy. The results of her recent quadruple screen are listed below: AFP: Low hCG: Low Estriol: Low Inhibin-A: Normal Which of the following is the most appropriate next step to confirm the diagnosis?
A 25-year-old man with a genetic disorder presents for genetic counseling because he is concerned about the risk that any children he has will have the same disease as himself. Specifically, since childhood he has had difficulty breathing requiring bronchodilators, inhaled corticosteroids, and chest physiotherapy. He has also had diarrhea and malabsorption requiring enzyme replacement therapy. If his wife comes from a population where 1 in 10,000 people are affected by this same disorder, which of the following best represents the likelihood a child would be affected as well?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
