Respiratory — MCQs

On this page
A 30-year-old woman presents to the emergency department with breathlessness for the last hour. She is unable to provide any history due to her dyspnea. Her vitals include: respiratory rate 20/min, pulse 100/min, and blood pressure 144/84 mm Hg. On physical examination, she is visibly obese, and her breathing is labored. There are decreased breath sounds and hyperresonance to percussion across all lung fields bilaterally. An arterial blood gas is drawn, and the patient is placed on inhaled oxygen. Laboratory findings reveal: pH 7.34 pO2 63 mm Hg pCO2 50 mm Hg HCO3 22 mEq/L Her alveolar partial pressure of oxygen is 70 mm Hg. Which of the following is the most likely etiology of this patient’s symptoms?
An investigator studying new drug delivery systems administers an aerosol containing 6.7-μm sized particles to a healthy subject via a nonrebreather mask. Which of the following is the most likely route of clearance of the particulate matter in this subject?
A 71-year-old man is admitted to the ICU with a history of severe pancreatitis and new onset difficulty breathing. His vital signs are a blood pressure of 100/60 mm Hg, heart rate of 100/min, respirations of 27/min, temperature of 36.7°C (98.1°F), and oxygen saturation of 85% on room air. Physical examination shows a cachectic male in severe respiratory distress. Rales are heard at the base of each lung. The patient is intubated and a Swan-Ganz catheter is inserted. Pulmonary capillary wedge pressure is 8 mm Hg. An arterial blood gas study reveals a PaO2: FiO2 ratio of 180. The patient is diagnosed with acute respiratory distress syndrome. In which of the following segments of the respiratory tract are the cells responsible for the symptoms observed in this patient found?
A 21-year-old lacrosse player comes to the doctor for an annual health assessment. She does not smoke or drink alcohol. She is 160 cm (5 ft 3 in) tall and weighs 57 kg (125 lb); BMI is 22 kg/m2. Pulmonary function tests show an FEV1 of 90% and an FVC of 3600 mL. Whole body plethysmography is performed to measure airway resistance. Which of the following structures of the respiratory tree is likely to have the highest contribution to total airway resistance?
A 64-year-old man presents to his primary care physician for follow-up of a severe, unrelenting, productive cough of 2 years duration. The medical history includes type 2 diabetes mellitus, which is well-controlled with insulin. He has a 25-pack-year smoking history and is an active smoker. The blood pressure is 135/88 mm Hg, the pulse is 94/min, the temperature is 36.9°C (98.5°F), and the respiratory rate is 18/min. Bilateral wheezes and crackles are heard on auscultation. A chest X-ray reveals cardiomegaly, increased lung markings, and a flattened diaphragm. Which of the following is most likely in this patient?
A 21-year-old man is admitted to the intensive care unit for respiratory failure requiring mechanical ventilation. His minute ventilation is calculated to be 7.0 L/min, and his alveolar ventilation is calculated to be 5.1 L/min. Which of the following is most likely to decrease the difference between minute ventilation and alveolar ventilation?
A 34-year-old woman comes to a physician for a routine health maintenance examination. She moved to Denver 1 week ago after having lived in New York City all her life. She has no history of serious illness and takes no medications. Which of the following sets of changes is most likely on analysis of a blood sample obtained now compared to prior to her move? Erythropoietin level | O2 saturation | Plasma volume
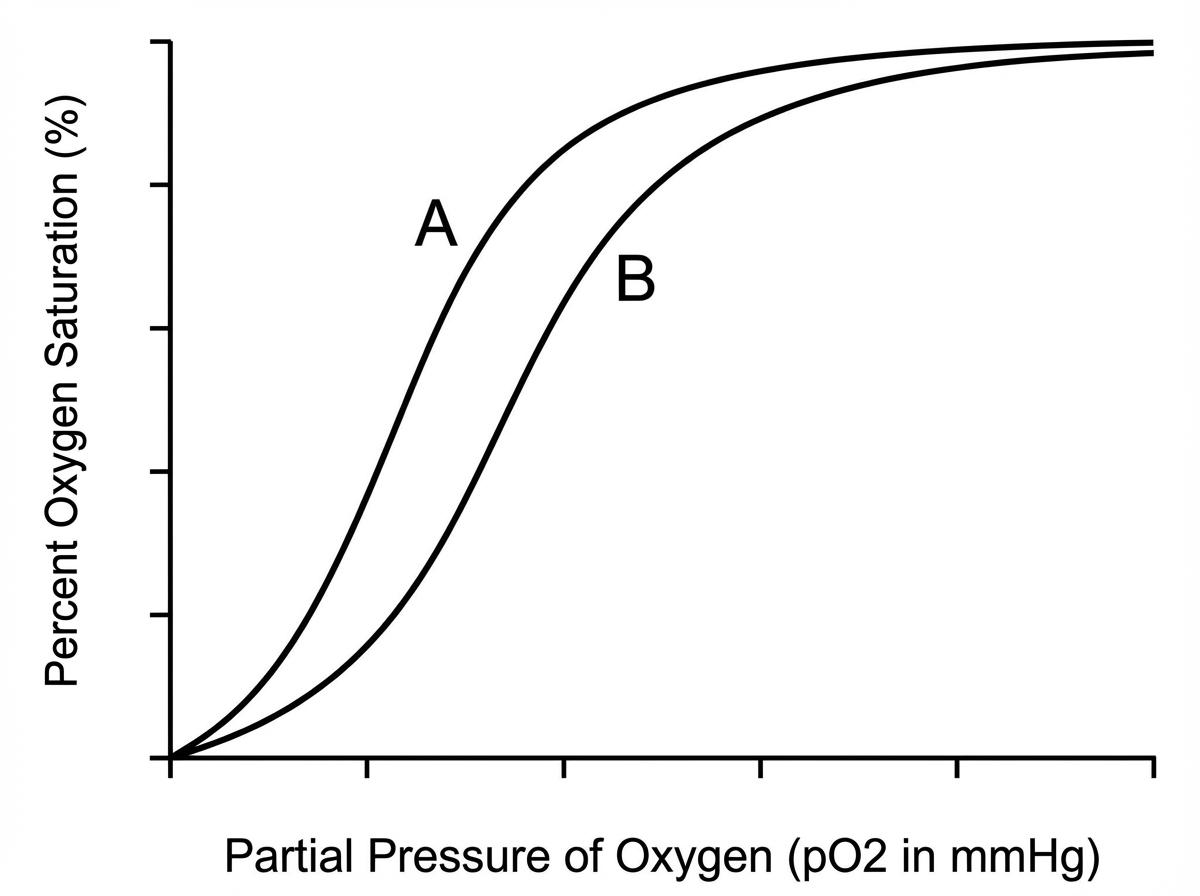
An investigator is conducting a study on hematological factors that affect the affinity of hemoglobin for oxygen. An illustration of two graphs (A and B) that represent the affinity of hemoglobin for oxygen is shown. Which of the following best explains a shift from A to B?
Which of the following physiologic changes decreases pulmonary vascular resistance (PVR)?
A 10-year-old boy is brought to the clinic by his mother with complaints of cough productive of yellow sputum for the past couple of weeks. This is the 4th episode the boy has had this year. He has had recurrent episodes of cough since childhood, and previous episodes have subsided with antibiotics. There is no family history of respiratory disorders. His vaccinations are up to date. He has a heart rate of 98/min, respiratory rate of 13/min, temperature of 37.6°C (99.7°F), and blood pressure of 102/70 mm Hg. Auscultation of the chest reveals an apex beat on the right side of the chest. A chest X-ray reveals that the cardiac apex is on the right. A high-resolution CT scan is performed which is suggestive of bronchiectasis. Which of the following structures is most likely impaired in this patient?
Practice by Chapter
Respiratory mechanics and work of breathing
Practice Questions
Pulmonary volumes and capacities
Practice Questions
Spirometry interpretation
Practice Questions
Flow-volume loops
Practice Questions
Airway resistance determinants
Practice Questions
Control of breathing
Practice Questions
Central and peripheral chemoreceptors
Practice Questions
Respiratory centers in the brainstem
Practice Questions
Exercise respiratory physiology
Practice Questions
Sleep effects on respiration
Practice Questions
High altitude adaptation
Practice Questions
Dyspnea mechanisms
Practice Questions
Integrated respiratory responses
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
