Titratable acid excretion — MCQs

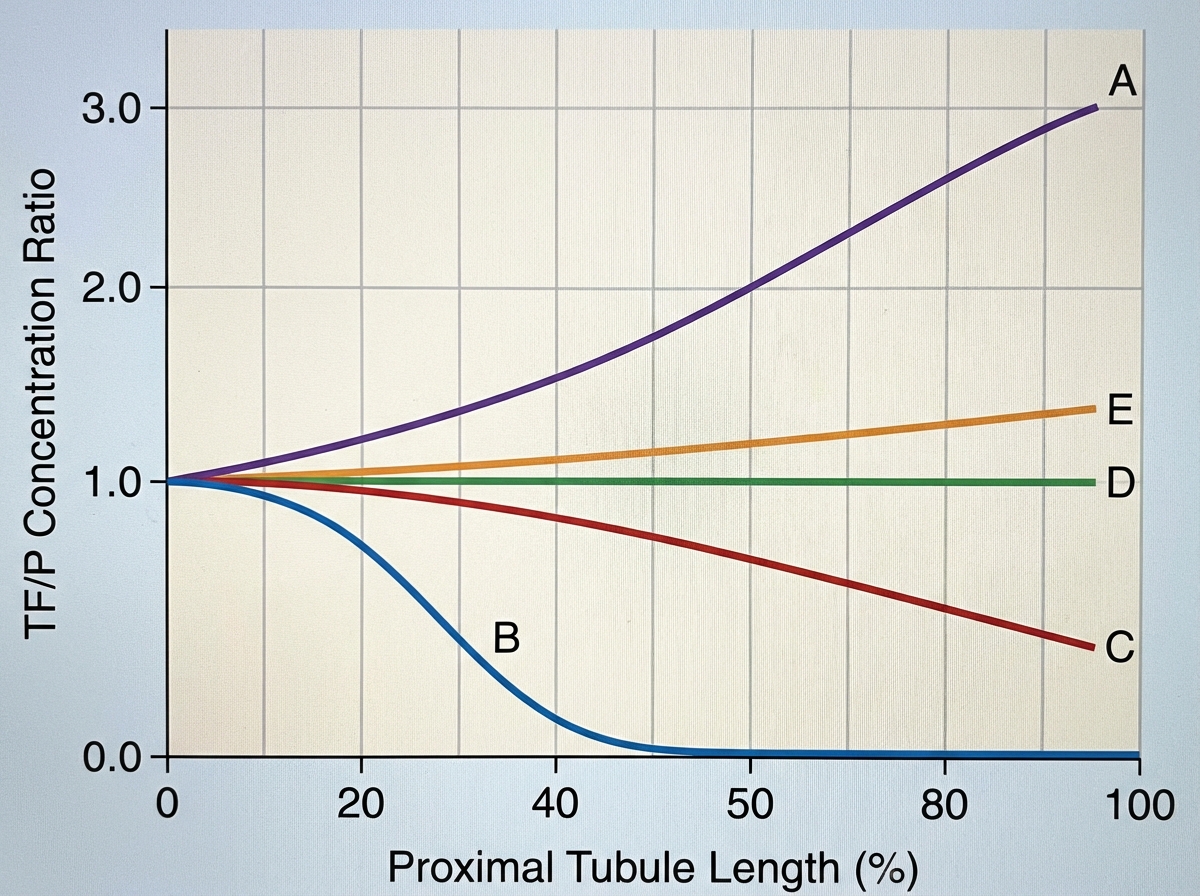
A 72-year-old female is brought to the emergency department after being found unresponsive in her garage with an open bottle of unmarked fluid. She is confused and is unable to answer questions on arrival. Her medical history is significant for Alzheimer disease, but her family says she has no medical comorbidities. Serum analysis of this patient's blood shows a pH of 7.28 with a high anion gap. The electrolyte that is most likely significantly decreased in this patient follows which of the following concentration curves across the proximal tubule of the kidney?
On cardiology service rounds, your team sees a patient admitted with an acute congestive heart failure exacerbation. In congestive heart failure, decreased cardiac function leads to decreased renal perfusion, which eventually leads to excess volume retention. To test your knowledge of physiology, your attending asks you which segment of the nephron is responsible for the majority of water absorption. Which of the following is a correct pairing of the segment of the nephron that reabsorbs the majority of all filtered water with the means by which that segment absorbs water?
A 45-year-old woman presents with severe, acute-onset colicky abdominal pain and nausea. She also describes bone pain, constipation, headache, decreased vision, and menstrual irregularity. Past medical history is significant for surgical removal of an insulinoma one year ago. Two months ago, she was prescribed fluoxetine for depression but hasn’t found it very helpful. Family history is significant for a rare genetic syndrome. Non-contrast CT, CBC, CMP, and urinalysis are ordered in the diagnostic work-up. Urine sediment is significant for the findings shown in the picture. Which of the following will also be a likely significant finding in the diagnostic workup?

In a healthy patient with no renal abnormalities, several mechanisms are responsible for moving various filtered substances into and out of the tubules. Para-aminohippurate (PAH) is frequently used to estimate renal blood flow when maintained at low plasma concentrations. The following table illustrates the effect of changing plasma PAH concentrations on PAH excretion: Plasma PAH concentration (mg/dL) | Urinary PAH concentration (mg/dL) 0 | 0 10 | 60 20 | 120 30 | 150 40 | 180 Which of the following mechanisms best explains the decreased rate of increase in PAH excretion observed when plasma PAH concentration exceeds 20 mg/dL?
A 72-year-old man being treated for benign prostatic hyperplasia (BPH) is admitted to the emergency department for 1 week of dysuria, nocturia, urge incontinence, and difficulty initiating micturition. His medical history is relevant for hypertension, active tobacco use, chronic obstructive pulmonary disease, and BPH with multiple urinary tract infections. Upon admission, he is found with a heart rate of 130/min, respiratory rate of 19/min, body temperature of 39.0°C (102.2°F), and blood pressure of 80/50 mm Hg. Additional findings during the physical examination include decreased breath sounds, wheezes, crackles at the lung bases, and intense right flank pain. A complete blood count shows leukocytosis and neutrophilia with a left shift. A sample for arterial blood gas analysis (ABG) was taken, which is shown below. Laboratory test Serum Na+ 140 mEq/L Serum Cl- 102 mEq/L Serum K+ 4.8 mEq/L Serum creatinine (SCr) 2.3 mg/dL Arterial blood gas pH 7.12 Po2 82 mm Hg Pco2 60 mm Hg SO2% 92% HCO3- 12.0 mEq/L Which of the following best explains the patient’s condition?
A 25-year-old woman with an extensive psychiatric history is suspected of having metabolic acidosis after ingesting a large amount of aspirin in a suicide attempt. Labs are drawn and the values from the ABG are found to be: PCO2: 25, and HCO3: 15, but the pH value is smeared on the print-out and illegible. The medical student is given the task of calculating the pH using the pCO2 and HCO3 concentrations. He recalls from his first-year physiology course that the pKa of relevance for the bicarbonate buffering system is approximately 6.1. Which of the following is the correct formula the student should use, using the given values from the incomplete ABG?
A 70-year-old woman is brought to the emergency department due to worsening lethargy. She lives with her husband who says she has had severe diarrhea for the past few days. Examination shows a blood pressure of 85/60 mm Hg, pulse of 100/min, and temperature of 37.8°C (100.0°F). The patient is stuporous, while her skin appears dry and lacks turgor. Laboratory tests reveal: Serum electrolytes Sodium 144 mEq/L Potassium 3.5 mEq/L Chloride 115 mEq/L Bicarbonate 19 mEq/L Serum pH 7.3 PaO2 80 mm Hg Pco2 38 mm Hg This patient has which of the following acid-base disturbances?
A group of researchers wish to develop a clinical trial assessing the efficacy of a specific medication on the urinary excretion of amphetamines in intoxicated patients. They recruit 50 patients for the treatment arm and 50 patients for the control arm of the study. Demographics are fairly balanced between the two groups. The primary end points include (1) time to recovery of mental status, (2) baseline heart rate, (3) urinary pH, and (4) specific gravity. Which medication should they use in order to achieve a statistically significant result positively favoring the intervention?
A group of investigators is studying a drug to treat refractory angina pectoris. This drug works by selectively inhibiting the late influx of sodium ions into cardiac myocytes. At high doses, the drug also partially inhibits the degradation of fatty acids. Which of the following is the most likely effect of this drug?
A 21-year-old man presents to the emergency department with acute back pain. The pain began a few hours prior to presentation and is located on the left lower back. The pain is described to be “shock-like,” 9/10 in pain severity, and radiates to the left groin. His temperature is 98.6°F (37°C), blood pressure is 120/75 mmHg, pulse is 101/min, and respirations are 18/min. The patient appears uncomfortable and is mildly diaphoretic. There is costovertebral angle tenderness and genitourinary exam is unremarkable. A non-contrast computerized tomography (CT) scan of the abdomen and pelvis demonstrates an opaque lesion affecting the left ureter with mild hydronephrosis. Straining of the urine with urine crystal analysis is demonstrated. Which of the following amino acids is most likely poorly reabsorbed by this patient’s kidney?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
