Henderson-Hasselbalch equation — MCQs

Which region of the nephron reabsorbs the highest percentage of filtered bicarbonate?
In your peripheral tissues and lungs, carbonic anhydrase works to control the equilibrium between carbon dioxide and carbonic acid in order to maintain proper blood pH. Through which mechanism does carbonic anhydrase exert its influence on reaction kinetics?
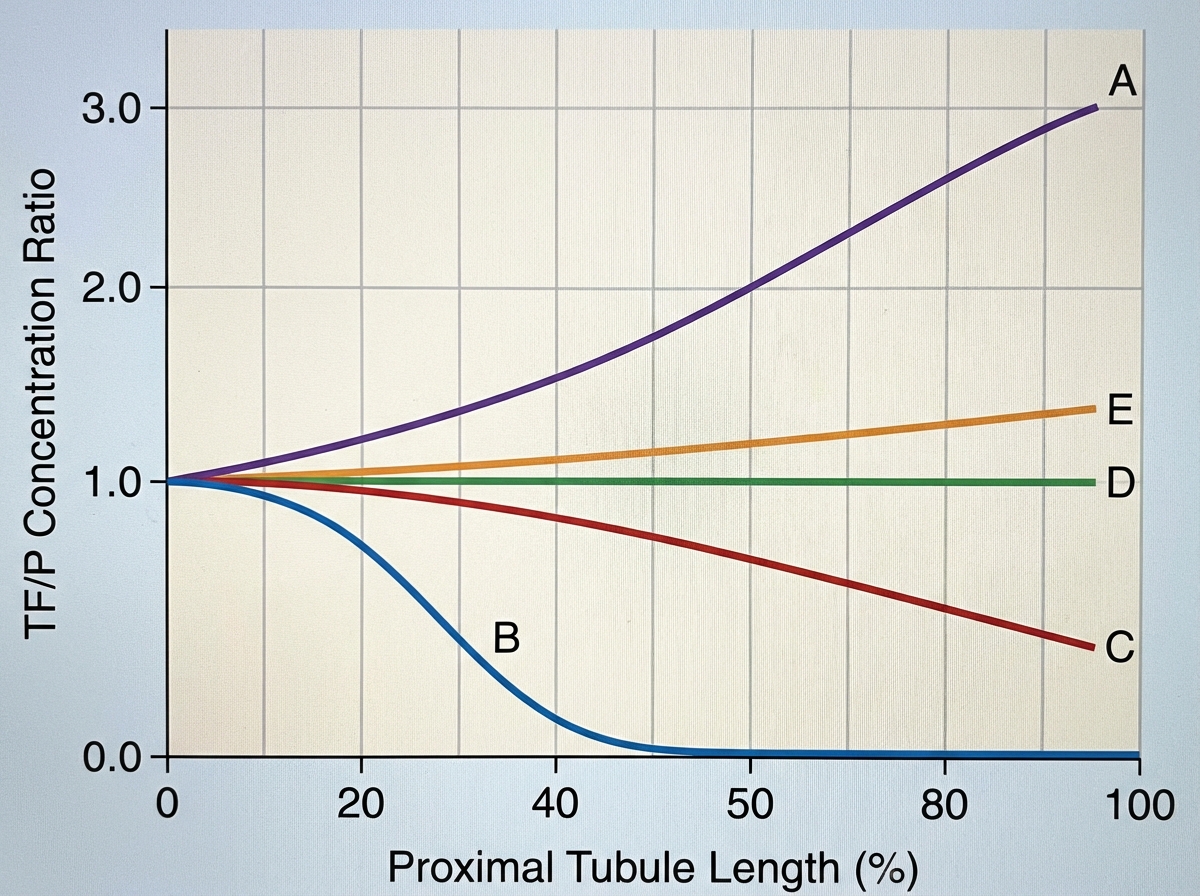
A 72-year-old female is brought to the emergency department after being found unresponsive in her garage with an open bottle of unmarked fluid. She is confused and is unable to answer questions on arrival. Her medical history is significant for Alzheimer disease, but her family says she has no medical comorbidities. Serum analysis of this patient's blood shows a pH of 7.28 with a high anion gap. The electrolyte that is most likely significantly decreased in this patient follows which of the following concentration curves across the proximal tubule of the kidney?
A 52-year-old man with a history of Type 1 diabetes mellitus presents to the emergency room with increasing fatigue. Two days ago, he ran out of insulin and has not had time to obtain a new prescription. He denies fevers or chills. His temperature is 37.2 degrees Celsius, blood pressure 84/56 mmHg, heart rate 100/min, respiratory rate 20/min, and SpO2 97% on room air. His physical exam is otherwise within normal limits. An arterial blood gas analysis shows the following: pH 7.25, PCO2 29, PO2 95, HCO3- 15. Which of the following acid-base disorders is present?
A 37-year-old G1P0 woman presents to her primary care physician for a routine checkup. She has a history of diabetes and hypertension but has otherwise been healthy with no change in her health status since the last visit. She is expecting her first child 8 weeks from now. She also enrolled in a study about pregnancy where serial metabolic panels and arterial blood gases are obtained. Partial results from these studies are shown below: Serum: Na+: 141 mEq/L Cl-: 108 mEq/L pH: 7.47 pCO2: 30 mmHg HCO3-: 21 mEq/L Which of the following disease processes would most likely present with a similar panel of metabolic results?
A 25-year-old woman with an extensive psychiatric history is suspected of having metabolic acidosis after ingesting a large amount of aspirin in a suicide attempt. Labs are drawn and the values from the ABG are found to be: PCO2: 25, and HCO3: 15, but the pH value is smeared on the print-out and illegible. The medical student is given the task of calculating the pH using the pCO2 and HCO3 concentrations. He recalls from his first-year physiology course that the pKa of relevance for the bicarbonate buffering system is approximately 6.1. Which of the following is the correct formula the student should use, using the given values from the incomplete ABG?
A 24-year-old male is brought in by ambulance to the emergency department after he was found unresponsive at home for an unknown length of time. Upon arrival, he is found to be severely altered and unable to answer questions about his medical history. Based on clinical suspicion, a panel of basic blood tests are obtained including an arterial blood gas, which shows a pH of 7.32, a pCO2 of 70, and a bicarbonate level of 30 mEq/L. Which of the following is most likely the primary disturbance leading to the values found in the ABG?
A 32-year-old female with Crohn's disease diagnosed in her early 20s comes to your office for a follow-up appointment. She is complaining of headaches and fatigue. Which of the following arterial blood gas findings might you expect?
A 44-year-old Caucasian male complains of carpopedal spasms, peri-oral numbness, and paresthesias of the hands and feet. His wife also mentions that he had a seizure not too long ago. His past surgical history is significant for total thyroidectomy due to papillary thyroid carcinoma. They then realized all of the symptoms occurred after the surgery. Which of the following would be present in this patient?
A 41-year-old man presents to urgent care with a 1-week history of severe diarrhea. He says that he has been having watery stools every 2-3 hours. The stools do not contain blood and do not float. On presentation, he is observed to have significant facial flushing, and laboratory tests reveal the following: Serum: Na+: 137 mEq/L K+: 2.7 mEq/L Cl-: 113 mEq/L HCO3-: 14 mEq/L A computed tomography scan reveals a small intra-abdominal mass. Staining of this mass would most likely reveal production of which of the following?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
