Acid-base balance — MCQs

On this page
A 44-year-old Caucasian male complains of carpopedal spasms, peri-oral numbness, and paresthesias of the hands and feet. His wife also mentions that he had a seizure not too long ago. His past surgical history is significant for total thyroidectomy due to papillary thyroid carcinoma. They then realized all of the symptoms occurred after the surgery. Which of the following would be present in this patient?
A 41-year-old man presents to urgent care with a 1-week history of severe diarrhea. He says that he has been having watery stools every 2-3 hours. The stools do not contain blood and do not float. On presentation, he is observed to have significant facial flushing, and laboratory tests reveal the following: Serum: Na+: 137 mEq/L K+: 2.7 mEq/L Cl-: 113 mEq/L HCO3-: 14 mEq/L A computed tomography scan reveals a small intra-abdominal mass. Staining of this mass would most likely reveal production of which of the following?
A 67-year-old woman presents to the Emergency Department complaining of weakness and fatigue. She says she caught a “stomach bug” and has not been able to eat anything without vomiting for three days. Past medical history is significant for hyperlipidemia. She takes atorvastatin and a multivitamin daily, except for the last two days due to nausea. Today her heart rate is 106/min, respiratory rate is 16/min, temperature is 37.6°C (99.7°F) and blood pressure of 110/70 mm Hg. On physical examination, her oral mucosa is dry and she looks pale and uncomfortable. She is admitted for care and administered ondansetron. An intravenous infusion of normal saline is also initiated. An arterial blood gas is collected. Which of the following results is expected to be seen in this patient?
A 22-year-old woman presents to the physician due to lightheadedness. Earlier in the day, she had her first job interview since graduating from college 3 months ago. While waiting outside the interviewer’s office, she began to feel nervous and started breathing really fast. She then felt as if she was going to faint. She excused herself from the interview, and requested a friend to drive her to the clinic. Which of the following is responsible for her symptoms?
A 54-year-old man presents with 3 days of non-bloody and non-bilious emesis every time he eats or drinks. He has become progressively weaker and the emesis has not improved. He denies diarrhea, fever, or chills and thinks his symptoms may be related to a recent event that involved sampling many different foods. His temperature is 97.5°F (36.4°C), blood pressure is 133/82 mmHg, pulse is 105/min, respirations are 15/min, and oxygen saturation is 98% on room air. Physical exam is notable for a weak appearing man with dry mucous membranes. His abdomen is nontender. Which of the following laboratory changes would most likely be seen in this patient?
A 56-year-old woman with a longstanding history of gastroesophageal reflux presents for follow-up evaluation of endoscopically confirmed gastric and duodenal ulcers. Her symptoms have been unresponsive to proton pump inhibitors and histamine receptor antagonists in the past. Results for H. pylori infection are still pending. Which of the following changes is expected in the patient's duodenum, given her peptic ulcer disease?
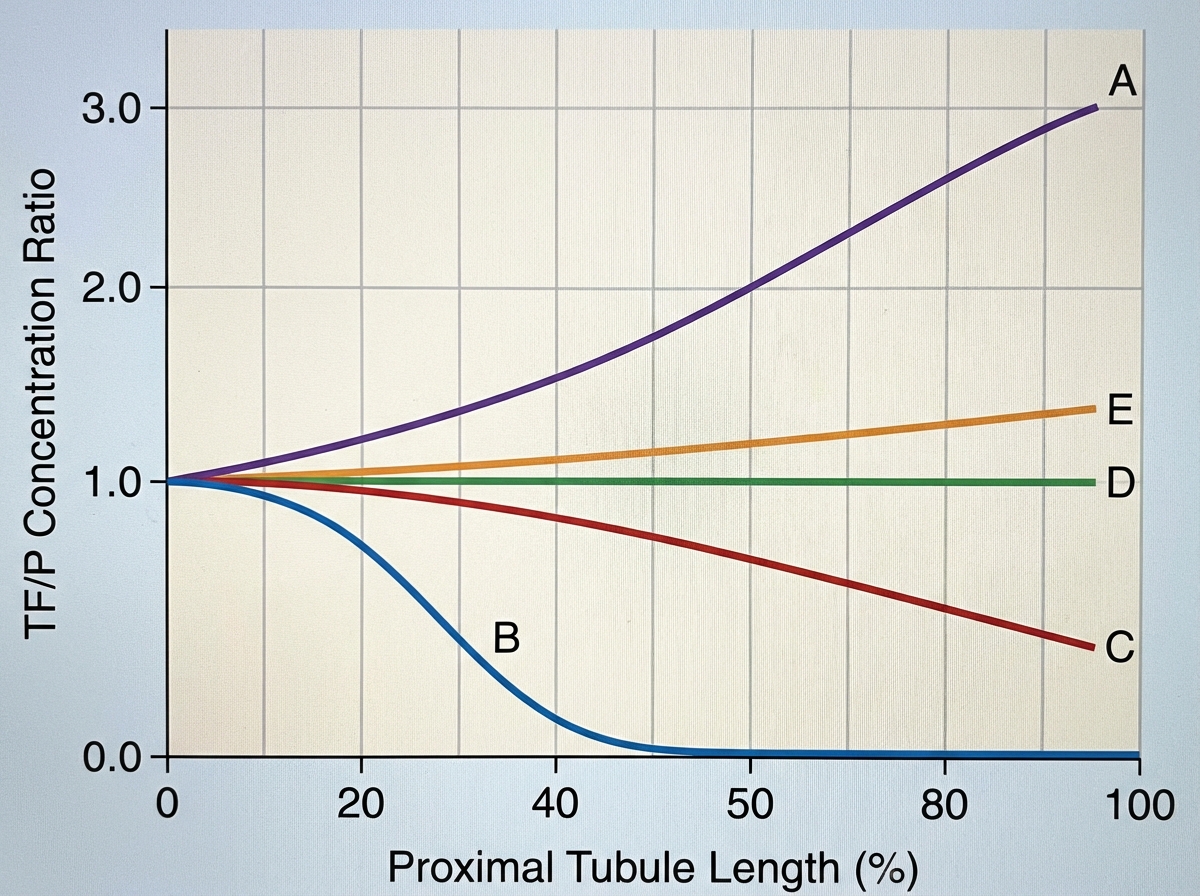
A 72-year-old female is brought to the emergency department after being found unresponsive in her garage with an open bottle of unmarked fluid. She is confused and is unable to answer questions on arrival. Her medical history is significant for Alzheimer disease, but her family says she has no medical comorbidities. Serum analysis of this patient's blood shows a pH of 7.28 with a high anion gap. The electrolyte that is most likely significantly decreased in this patient follows which of the following concentration curves across the proximal tubule of the kidney?
A healthy 20-year-old male college student attempts to climb Mount Everest and travels to the Tibetan plateau by plane. Upon landing, he feels increasingly dizzy and fatigued. He notices that he is breathing faster than usual. What is the initial stimulus for the most likely acid-base disorder?
A 32-year-old man is brought to the emergency department after he was found unresponsive on the street. Upon admission, he is lethargic and cyanotic with small, symmetrical pinpoint pupils. The following vital signs were registered: blood pressure of 100/60 mm Hg, heart rate of 70/min, respiratory rate of 8/min, and a body temperature of 36.0°C (96.8°F). While being assessed and resuscitated, a sample for arterial blood gas (ABG) analysis was taken, in addition to the following biochemistry tests: Laboratory test Serum Na+ 138 mEq/L Serum Cl- 101 mEq/L Serum K+ 4.0 mEq/L Serum creatinine (SCr) 0.58 mg/dL Which of the following values would you most likely expect to see in this patient’s ABG results?
A 67-year-old man presents to his primary care physician because of weak urine stream, and increasing difficulty in initiating and stopping urination. He also reports of mild generalized body aches and weakness during the day. The past medical history includes diabetes mellitus type 2 for 35 years and essential hypertension for 19 years. The medication list includes metformin, vildagliptin, and enalapril. The vital signs include: temperature 36.7°C (98.1°F), blood pressure 151/82 mm Hg, and pulse 88/min. The physical examination is remarkable for markedly enlarged, firm prostate without nodules. The laboratory test results are as follows: Serum sodium 142 mEq/L Serum potassium 5.7 mEq/L Serum chloride 115 mEq/L Serum bicarbonate 17 mEq/L Serum creatinine 0.9 mg/dL Arterial pH 7.31 Urine pH 5.3 Urine sodium 59 mEq/L Urine potassium 6.2 mEq/L Urine chloride 65 mEq/L Which of the following most likely explains the patient’s findings?
Practice by Chapter
Buffer systems (bicarbonate, phosphate, protein)
Practice Questions
Henderson-Hasselbalch equation
Practice Questions
Renal regulation of acid-base balance
Practice Questions
Renal bicarbonate handling
Practice Questions
Ammonia production and excretion
Practice Questions
Titratable acid excretion
Practice Questions
Respiratory regulation of acid-base
Practice Questions
Metabolic acidosis mechanisms and compensation
Practice Questions
Metabolic alkalosis mechanisms and compensation
Practice Questions
Respiratory acidosis mechanisms and compensation
Practice Questions
Respiratory alkalosis mechanisms and compensation
Practice Questions
Mixed acid-base disorders
Practice Questions
Clinical approach to acid-base disorders
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
