Antivirals — MCQs

On this page
A 65-year-old patient presents with symptoms of bone pain, anemia, hypercalcemia, and renal impairment. A bone marrow biopsy confirms the diagnosis of multiple myeloma. The patient is started on a treatment regimen. Which of the following treatments is most likely associated with the reactivation of herpes zoster?
A patient with HIV who is currently on antiretroviral therapy consisting of zidovudine, lamivudine, and nevirapine is diagnosed with tuberculosis. Considering potential drug interactions, which of the following TB drugs should be changed in this patient?
A 28-year-old primigravid woman at 38 weeks' gestation is brought to the emergency department in active labor. She has not had regular prenatal care. She has a history of HIV infection but is not currently on antiretroviral therapy. Her previous viral load is unknown. Treatment with intravenous zidovudine is begun to reduce perinatal transmission of the virus. Which of the following processes is most likely affected by this drug?
The physician recommends that the patient receive an influenza vaccine. The patient becomes nervous and reports that he has never received an influenza vaccination because of an allergy to eggs. The allergy was diagnosed many years ago, after he developed hives upon eating scrambled eggs. Which of the following is the most appropriate next step in management?
A 35-year-old male is found to be infected with an HIV strain resistant to saquinavir and zidovudine. Which of the following best explains the drug resistance observed in this patient?
A 72-year-old man presents to the emergency department with a change in his behavior. The patient is brought in by his family who state that he is not acting normally and that his responses to their questions do not make sense. The patient has a past medical history of diabetes and Alzheimer dementia. His temperature is 103°F (39.4°C), blood pressure is 157/98 mmHg, pulse is 120/min, respirations are 19/min, and oxygen saturation is 98% on room air. Physical exam reveals a systolic murmur heard along the right upper sternal border. HEENT exam reveals a normal range of motion of the neck in all 4 directions and no lymphadenopathy. A mental status exam reveals a confused patient who is unable to answer questions. Laboratory values are ordered and a lumbar puncture is performed which demonstrates elevated white blood cells with a lymphocytic predominance, a normal glucose, and an elevated protein. The patient is started on IV fluids and ibuprofen. Which of the following is the next best step in management?
A 44-year-old man comes to the physician for a follow-up examination. Ten months ago, he was diagnosed with HIV infection and appropriate antiretroviral therapy was initiated. Physical examination shows no abnormalities. Laboratory studies show increased viral load despite ongoing treatment. His pharmacotherapy is switched to a new combination drug regimen including an agent that binds to glycoprotein 41. The expected effect of this drug is most likely due to inhibition of which of the following?
A 55-year-old man with HIV on antiretroviral therapy comes to the physician for a follow-up examination. His HIV viral load is 559 copies/mL (N<49). His physician is concerned about the development of drug resistance. The result of HIV genotype testing shows reduced viral susceptibility to darunavir and ritonavir. Which of the following molecular processes is most likely affected by this mutation?
A 63-year-old HIV-positive man comes to the physician for a routine health maintenance examination. Four years ago, he was diagnosed with HIV and was started on cART therapy. He tells the physician that he has been having difficulty adhering to his medication regimen. He has been unemployed for the past couple of years and relies on unemployment benefits to cover the costs of daily living. His father died of lymphoma at the age of 60 years. He wants more information about his risk of developing DLBCL. Which of the following is the greatest risk factor for the development of DLBCL in HIV-positive patients?
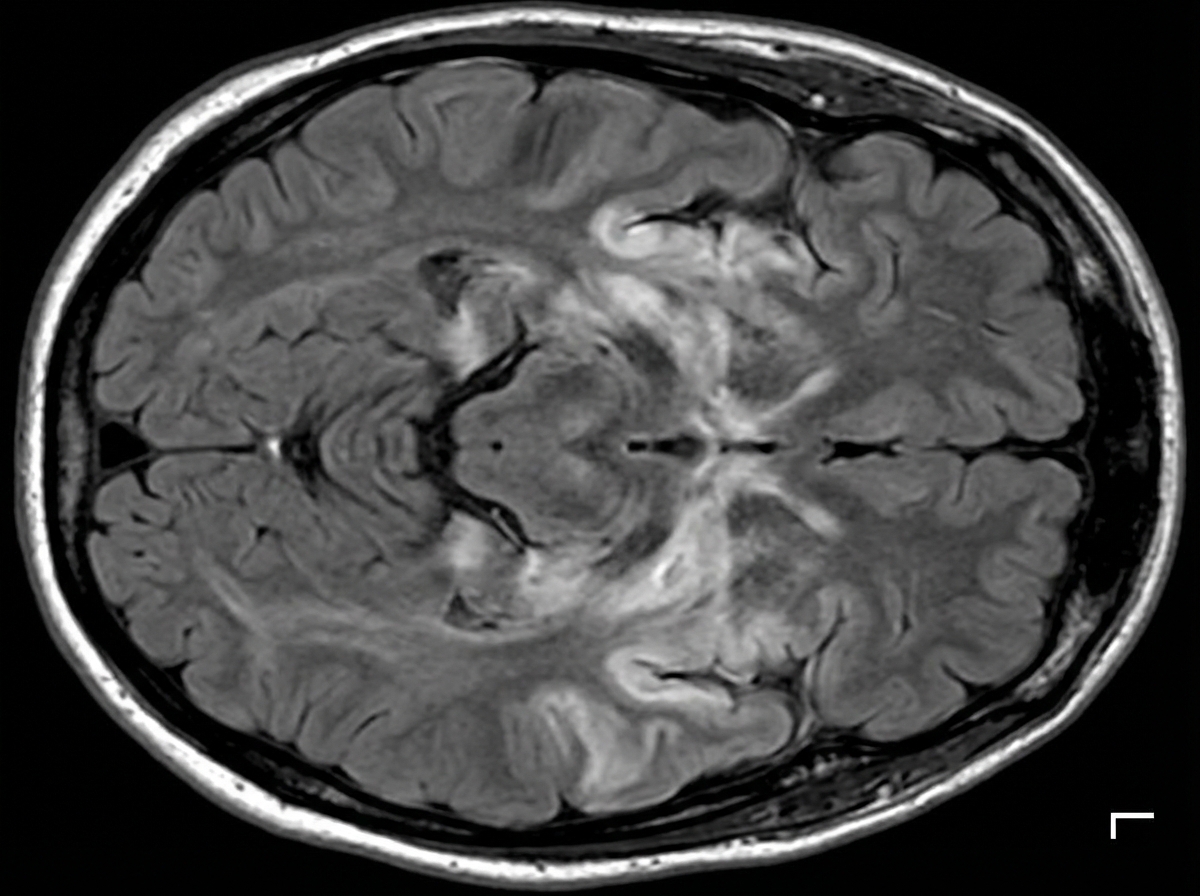
A 27-year-old man is brought to the emergency department by his friends in a confused state. He was doing fine 5 days ago when he started to complain of fever and flu-like symptoms. His fever was low-grade and associated with a headache. For the past 2 days, he has become increasingly irritable, confused, and was getting angry at trivial things. Past medical history is unremarkable. He is a college student and is physically active. He smokes cigarettes occasionally. He drinks alcohol socially. He is sexually active with his girlfriend and they use condoms inconsistently. Physical examination reveals: blood pressure 120/80 mm Hg, heart rate 108/min, respiratory rate 10/min, and temperature 37.4°C (99.4°F). He is confused and disoriented. Pupils are 3 mm in diameter and respond to light sluggishly. He is moving all his limbs spontaneously. His neck is supple. MRI of the brain is shown in the picture. Cerebrospinal fluid (CSF) reveals an opening pressure of 16 cm of H20, a total leukocyte count of 112/mm3 with 85% lymphocytes, the protein of 42 mg/dL, and glucose of 58 mg/dL. What is the best treatment for this condition?
Practice by Chapter
HIV protease inhibitors
Practice Questions
HIV reverse transcriptase inhibitors
Practice Questions
HIV integrase inhibitors
Practice Questions
HIV entry inhibitors
Practice Questions
Hepatitis B antivirals
Practice Questions
Hepatitis C direct-acting antivirals
Practice Questions
Influenza antivirals
Practice Questions
Herpesvirus antivirals
Practice Questions
Cytomegalovirus antivirals
Practice Questions
Respiratory syncytial virus therapies
Practice Questions
Broad-spectrum antivirals
Practice Questions
Antiretroviral resistance
Practice Questions
Antiviral prophylaxis strategies
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
