Neoplasia — MCQs

On this page
A 71-year-old African American man with a history of prostatic adenocarcinoma presents to his oncologist with low back pain. He was diagnosed with non-resectable prostatic adenocarcinoma 4 years ago. He has undergone radiation therapy and chemotherapy. Over the past 3 months, he has developed constant mild non-radiating low back pain that occasionally wakes him up from sleep. He denies any recent falls or trauma. His past medical history is notable for hypertension, diabetes mellitus, coronary artery disease, and gout. He also has a history of thyroid cancer and underwent thyroidectomy 5 years ago. He takes lisinopril, metoprolol, aspirin, metformin, and allopurinol. He has a 40-pack-year smoking history and drinks alcohol socially. His temperature is 99.2°F (37.3°C), blood pressure is 150/85 mmHg, pulse is 84/min, and respirations are 18/min. On exam, he is well-appearing and in no acute distress. He is mildly tender to palpation along the lumbar vertebral spinous processes. A computerized tomography (CT) scan of the lumbar spine demonstrates a blastic and sclerotic lesion in the L5 vertebral body. Which of the following findings would most likely be seen on a serum study of this patient?
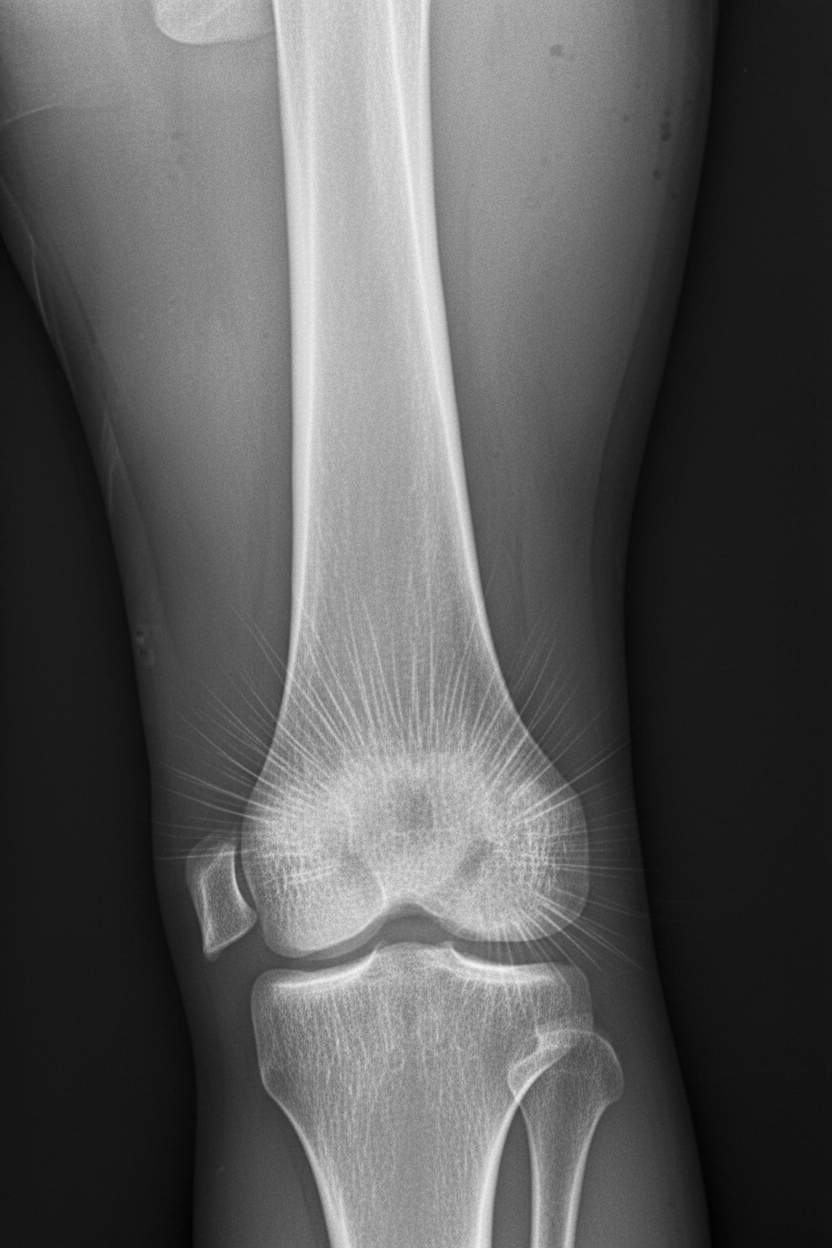
A 13-year-old boy is brought to his pediatrician for evaluation of leg pain. Specifically, he has been having pain around his right knee that has gotten progressively worse over the last several months. On presentation, he has swelling and tenderness over his right distal femur. Radiographs are obtained and the results are shown in figure A. His family history is significant in that several family members also had this disorder and others had pathology in the eye near birth. The patient is referred for a genetic consult, and a mutation is found on a certain chromosome. The chromosome that is most likely affected also contains a gene that is associated with which of the following pathologies?
A 32-year-old woman visits her primary care provider with the results of a recent colonoscopy, which was ordered after 3 episodes of rectal bleeding in the last month. Her grandmother, mother, and sister all have been diagnosed with nonpolyposis colorectal cancer, at ages 65, 50, and 40 years, respectively. Colonoscopy for this patient revealed a large, flat, right-sided adenoma. Histopathological examination of the lesion showed villous histology and high-grade dysplasia. Which of the following helps explain the condition of this patient?
A 58-year-old male undergoes a surveillance colonoscopy in which a 2 cm adenoma is identified and removed. Had this adenoma not been excised, the patient would have been at risk of progression to carcinoma. Which of the following is the final mutational step in the progression from adenoma to carcinoma?
A 16-year-old male presents to the emergency department complaining of episodes of pounding headache, chest fluttering, and excessive sweating. He has a past history of kidney stones that are composed of calcium oxalate. He does not smoke or drink alcohol. Family history reveals that his mother died of thyroid cancer. Vital signs reveal a temperature of 37.1°C (98.7°F), blood pressure of 200/110 mm Hg and pulse of 120/min. His 24-hour urine calcium, serum metanephrines, and serum normetanephrines levels are all elevated. Mutation of which of the following genes is responsible for this patient's condition?
A 38-year-old man presents to his primary care practitioner for 2 months of rectal bleeding. He also reports occasional diarrhea and abdominal pain. His family history is relevant for his father and uncle, who died from complications of colorectal cancer. Colonoscopy shows more than 10 colorectal adenomas. Which of the following genes is most likely affected in this patient?
A 72-year-old man presents to the emergency department with a complaint of rectal bleeding. He describes blood mixed in with the stool, which is associated with a change in his normal bowel habit such that he is going more frequently than normal. He also has some crampy left-sided abdominal pain and weight loss. His symptoms started 2 months ago, but he thought they are due to lack of dietary fiber intake and excess consumption of red meat. He has had type 2 diabetes mellitus for 35 years for which he takes metformin. He also uses daily low-dose aspirin for cardioprotection and occasional naproxen for knee pain. His family history is irrelevant. On examination, his abdomen and digital rectal examination are normal. Colonoscopy shows an ulcerating mucosal lesion with a narrow bowel lumen and biopsy shows a moderately differentiated adenocarcinoma. Which of the following is the greatest risk factor for colorectal cancer in this patient?
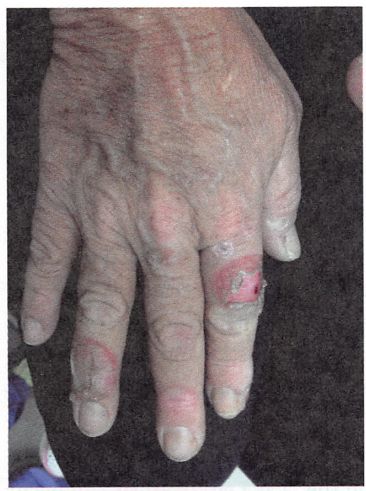
A 62-year-old man seeks evaluation at an outpatient clinic for a single, red, crusty lesion on the shaft of his penis and a similar lesion on the middle finger of his left hand. He recently immigrated to the US from Africa. The lesions are painless and the physicians in his country treated him for syphilis and eczema, with no improvement. He lives with his 4th wife. He smokes 2 packs of cigarette per day and has been doing so for the last 30 years. He is not aware of any family members with malignancies or hereditary diseases. The physical examination is remarkable for an erythematous plaque, with areas of crusting, oozing, and irregular borders on the dorsal surface of the penile shaft and a similar lesion on his left middle finger (shown in the picture). The regional lymph nodes are not affected. A biopsy is obtained and the pathologic evaluation reveals cells with nuclear hyperchromasia, multinucleation, and increased mitotic figures within the epidermis. What is the most likely diagnosis?
A 35-year-old woman, gravida 2, para 2, comes to the physician with intermenstrual bleeding and heavy menses for the past 4 months. She does not take any medications. Her father died of colon cancer at the age of 42 years. A curettage sample shows dysplastic tall, columnar, cells in the endometrium without intervening stroma. Germline sequencing shows a mutation in the MLH1 gene. Which of the following is the most likely underlying cause of neoplasia in this patient?
A 45-year-old woman presents with heavy menstrual bleeding between her periods. The patient also complains of experiencing an irregular menstrual cycle, weight loss, bloating, and constipation. She has had 3 uncomplicated pregnancies, all of which ended with normal vaginal deliveries at term. She has never taken oral contraception, and she does not take any medication at the time of presentation. She has no family history of any gynecological malignancy; however, her grandfather and mother had colon cancer that was diagnosed before they turned 50. On physical examination, the patient appears pale. Gynecological examination reveals a bloody cervical discharge and slight uterine enlargement. Endometrial biopsy reveals endometrial adenocarcinoma. Colonoscopy reveals several polyps located in the ascending colon, which are shown to be adenocarcinomas on histological evaluation. Which of the following mechanisms of DNA repair is likely to be disrupted in this patient?
Practice by Chapter
Characteristics of benign vs malignant tumors
Practice Questions
Nomenclature of neoplasms
Practice Questions
Carcinogenesis models
Practice Questions
Oncogenes and proto-oncogenes
Practice Questions
Tumor suppressor genes
Practice Questions
DNA repair genes and cancer
Practice Questions
Epigenetic mechanisms in cancer
Practice Questions
Apoptosis and cancer
Practice Questions
Tumor angiogenesis
Practice Questions
Tumor invasion and metastasis
Practice Questions
Carcinogenic agents
Practice Questions
Paraneoplastic syndromes
Practice Questions
Tumor immunology
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
