Neoplasia — MCQs

On this page
A 61-year-old Caucasian male presents to your office complaining of morning headaches of 6 weeks duration. A head MRI reveals a likely metastasis of unknown origin in the supratentorial region of the brain. On biopsy, the neoplastic mass is shown to have a mutation in BRAF, a protein kinase, in which a glutamic acid is substituted for valine at position 600 of the protein. Where did this metastasis most likely originate?
A 31-year-old woman comes to the emergency department because of a 4-week history of worsening headache, nausea, and vomiting. The headache is worse at night. Fundoscopic examination shows swelling of the optic discs. A CT scan of the brain shows a heterogeneous, hyperintense, intraventricular mass. The patient undergoes surgical excision of the mass. Pathologic examination of the surgical specimen confirms that the tumor is of neuronal origin. The cells in this specimen are most likely to stain positive for which of the following immunohistochemical markers?
A 67-year-old man comes to the physician because of a 4-month history of fatigue and weight loss. Physical examination shows jaundice. The liver is palpated 3 cm below the right costal margin. Serum studies show an elevated alpha-fetoprotein and a prolonged prothrombin time. Genetic analysis of a liver biopsy specimen shows a G:C to T:A transversion in codon 249 of the gene coding for the TP53 protein in affected cells. Which of the following risk factors is most specific to the patient's condition?
A 41-year-old construction worker presents to the office complaining of a progressively worsening breathlessness for the last 2 months. He has no other complaints. His medical history is significant for hypertension being treated with lisinopril-hydrochlorothiazide and gastroesophageal reflux disease being treated with pantoprazole. He has a 30-pack-year smoking history and drinks alcohol on the weekends. He works mainly with insulation and drywall placing. His temperature is 37.0°C (98.6°F), the blood pressure is 144/78 mm Hg, the pulse is 72/min, and the respirations are 10/min. Upon further questioning about his employment, the patient admits that he does not regularly use a mask or other protective devices at work. Which of the following malignancies is this patient most likely at risk for?
A 58-year-old woman with a history of breast cancer, coronary artery disease, gastroesophageal reflux, and diabetes mellitus is diagnosed with angiosarcoma. Which of the following most likely predisposed her to this condition?
A 56-year-old man presents to his primary care physician complaining of a dark spot on his left thigh. He says that he first noticed the spot about 3 years ago when he went to the beach with his family; however, at the time it was very small and he didn't think that it was worth mentioning. Since then, it has been growing slowly and he is now concerned about its size. He says that he does not have any other symptoms associated with the lesion. Physical exam reveals an asymmetric 2.5 centimeter plaque with irregular borders and a varying pattern of brown coloration. The lesion is studied and found to have a mutation in a tumor suppressor gene that normally prevents uncontrolled cell cycle progression by inhibiting cyclin-CDK complexes. Which of the following is most likely true of this gene?
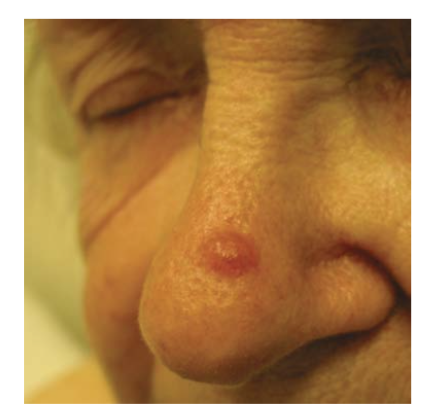
A 62-year-old man comes to the physician because of a skin lesion on his nose. The patient has had the lesion for 11 months and it has increased in size over the past few months. He is a farmer and lives together with his wife. His mother died of metastatic melanoma at the age of 67 years. The patient has smoked a pack of cigarettes daily for the past 30 years and drinks 1–2 glasses of whiskey on weekends. His temperature is 36.8°C (98.2°F), pulse is 75/min, and blood pressure is 140/78 mm Hg. Examination of the skin shows a nontender lesion at the right root of the nose. An image of the lesion is shown. Which of the following is the most likely diagnosis in this patient?
A 50-year-old man presents to his primary care provider complaining of smelling abnormal odors on several occasions. He says that he smells burnt rubber even though there is nothing burning and no one around him can smell what he does. This symptom has been intermittently bothering him for the past 6 months. Also during this period, he had occasional nosebleeds. He works as a high school teacher. Although his work gets a little stressful around the exam season, he says he is able to cope well. Family history is unremarkable. He does not smoke or drink alcohol and denies the use of any medication. Physical examination reveals unilateral nasal obstruction with some dried blood in the nasal passage. What is the most likely diagnosis?
A 57-year-old woman presents to the hospital complaining of 4 months of persistent abdominal pain and early satiety that has recently gotten worse. The patient says that she was prompted to come to the emergency department because she had several episodes of hematemesis. Her last menstrual period was approximately 8 years ago. The patient is sexually active with her husband and notes that she has recently had pain with intercourse as well as 'spotting' after intercourse. The patient states that she has also been experiencing nausea and weight loss associated with abdominal pain. Her blood pressure is 125/84 mm Hg, respiratory rate is 15/min, and heart rate is 76/min. Which of the following would be pathognomonic of this patient’s most likely diagnosis?
A 6-month-old boy is brought to the pediatrician for multiple swellings on his scalp. His mother reports that she first noticed 3 softened and swollen areas over the child's scalp 2 months ago that have grown in size. The child is also urinating more frequently than usual. He was born by cesarean section at 39 weeks gestation. The mother had appropriate prenatal care. She has a history of gastroesophageal reflux disease for which she takes omeprazole. Her family history is unknown as she was adopted at a young age. The boy's temperature is 99°F (37.2°C), blood pressure is 100/60 mmHg, pulse is 110/min, and respirations are 20/min. On exam, he has 3 areas of soft tissue swelling on his skull that are tender to palpation. Moderate asymmetric exophthalmos is noted. A water deprivation test is performed which demonstrates a urine specific gravity of 1.005. The urine specific gravity rises with desmopression administration. A head computerized tomography (CT) scan is performed which demonstrates multifocal lytic calvarial lesions. A biopsy of one of the lesions is performed. Analysis of the biopsy would most likely reveal which of the following findings?
Practice by Chapter
Characteristics of benign vs malignant tumors
Practice Questions
Nomenclature of neoplasms
Practice Questions
Carcinogenesis models
Practice Questions
Oncogenes and proto-oncogenes
Practice Questions
Tumor suppressor genes
Practice Questions
DNA repair genes and cancer
Practice Questions
Epigenetic mechanisms in cancer
Practice Questions
Apoptosis and cancer
Practice Questions
Tumor angiogenesis
Practice Questions
Tumor invasion and metastasis
Practice Questions
Carcinogenic agents
Practice Questions
Paraneoplastic syndromes
Practice Questions
Tumor immunology
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
