Hematopathology — MCQs

On this page
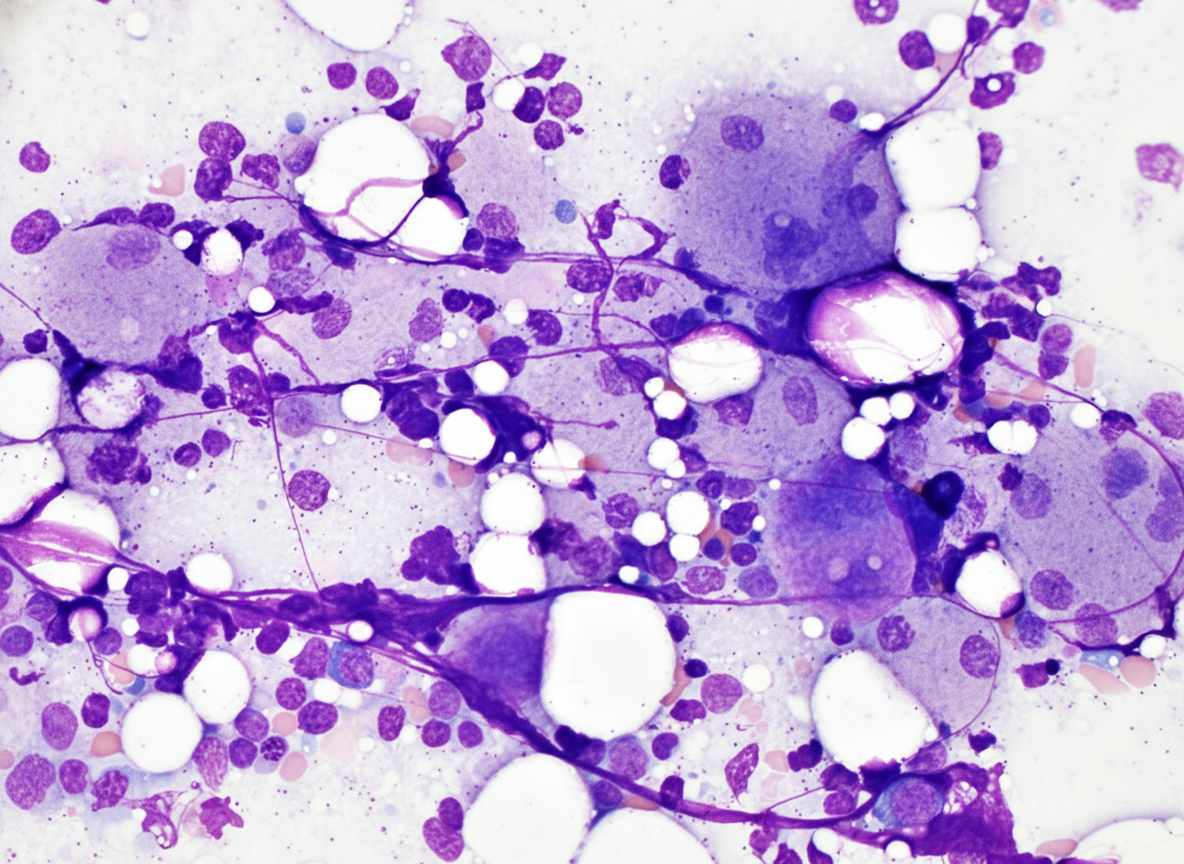
Infant with anemia, poor growth and bleeding from nose was brought to emergency. On examination hepatosplenomegaly was noted, and serum chitotriosidase levels are elevated. Bone marrow examination was done. Diagnosis is:
The vacutainer shown below is used for collecting sample for? (AIIMS Nov 2017)

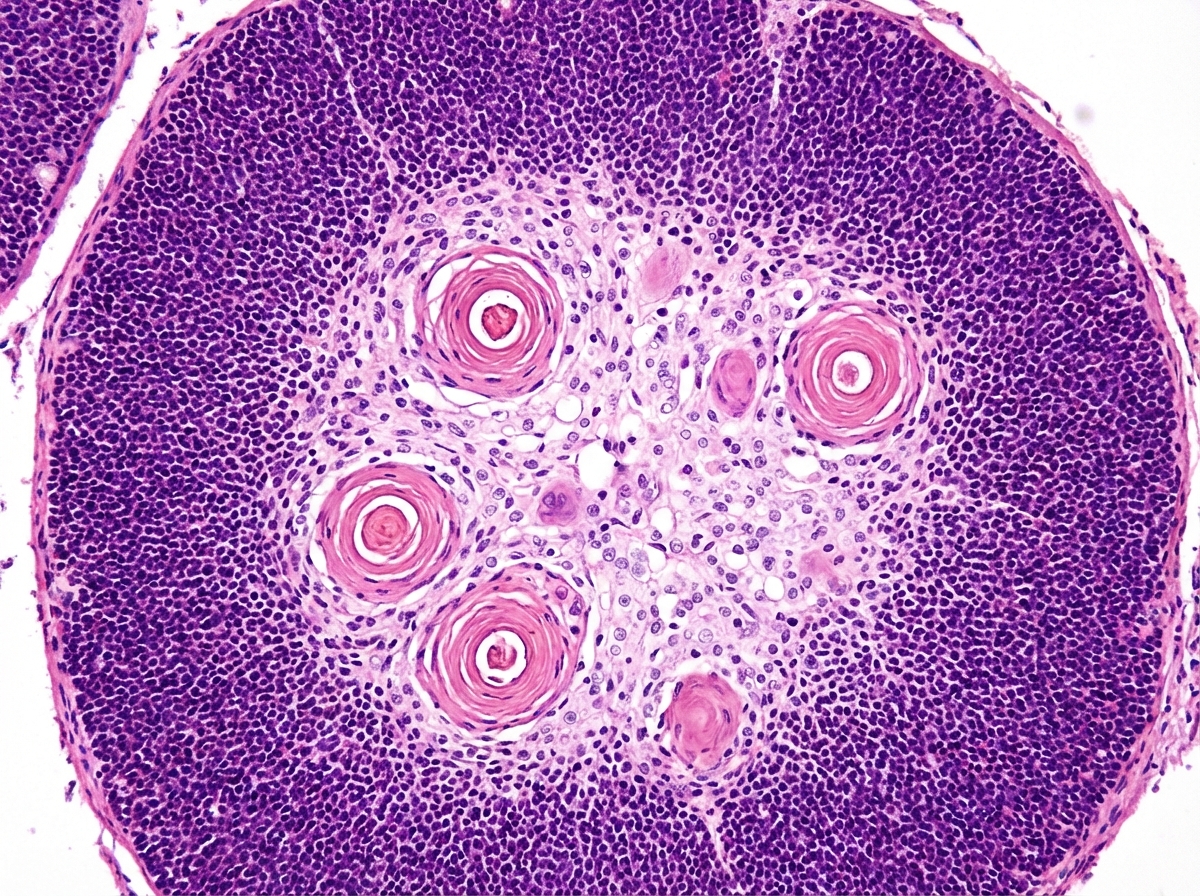
Identify the tissue:
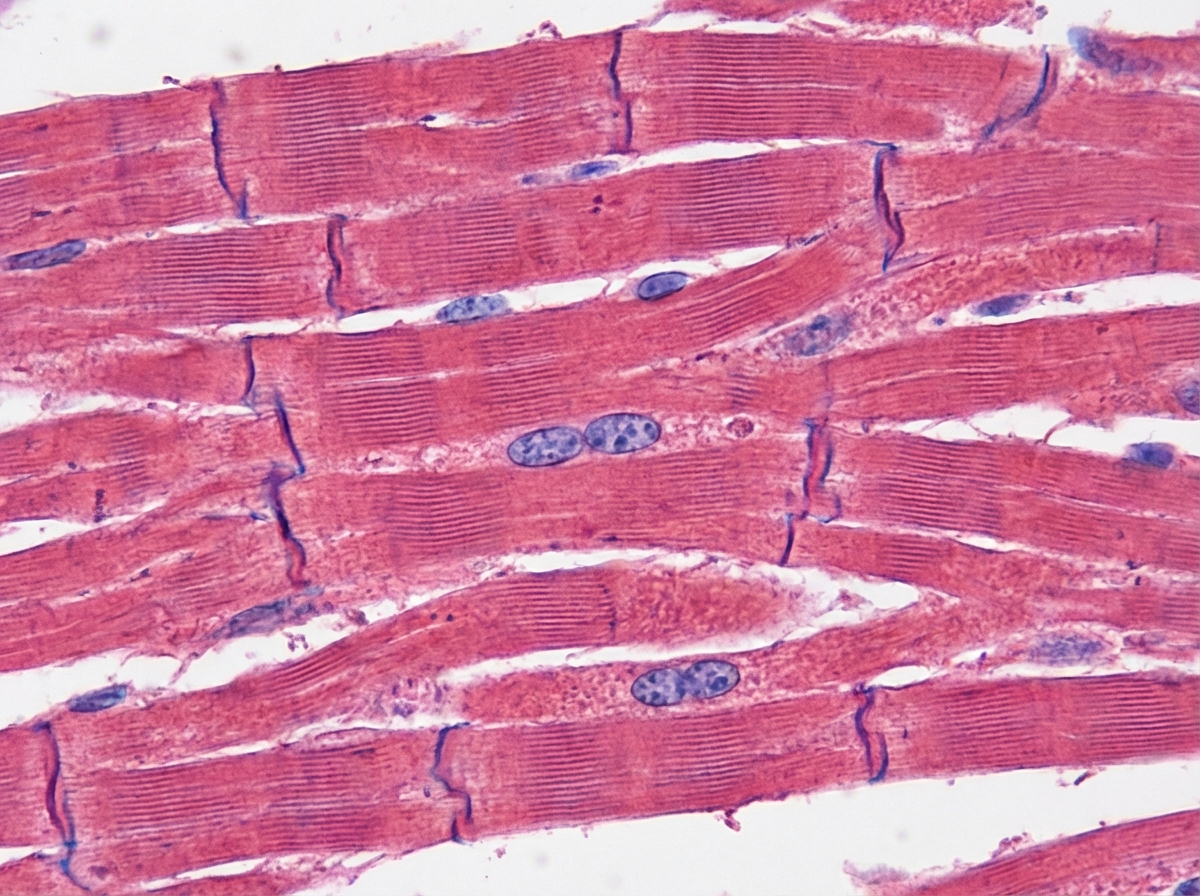
The image shows presence of:
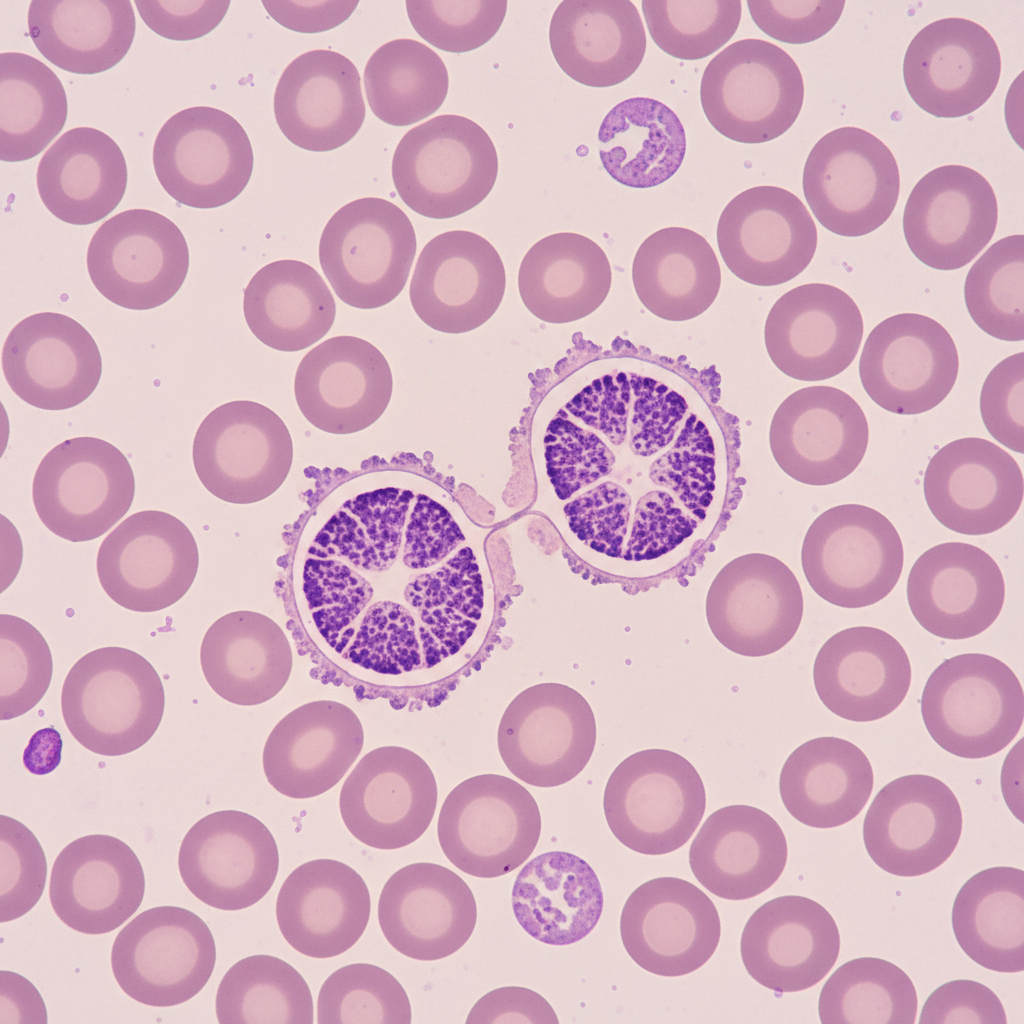
The image shows: (Recent NEET Pattern 2016-17)
Practice by Chapter
Red blood cell disorders
Practice Questions
White blood cell disorders
Practice Questions
Platelet disorders
Practice Questions
Coagulation disorders
Practice Questions
Acute leukemias
Practice Questions
Chronic leukemias
Practice Questions
Myeloproliferative neoplasms
Practice Questions
Myelodysplastic syndromes
Practice Questions
Hodgkin lymphoma
Practice Questions
Non-Hodgkin lymphomas
Practice Questions
Plasma cell disorders
Practice Questions
Bone marrow failure syndromes
Practice Questions
Splenic pathology
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
