Viruses — MCQs

On this page
A 27-year-old dental radiographer presented to a clinic with red lesions on his palate, right lower and mid-upper lip, as well as one of his fingers. These lesions were accompanied by slight pain, and the patient had a low-grade fever 1 week before the appearance of the lesions. The patient touched the affected area repeatedly, which resulted in bleeding. Two days prior to his visit, he observed a small vesicular eruption on his right index finger, which merged with other eruptions and became cloudy on the day of the visit. He has not had similar symptoms previously. He did not report drug usage. A Tzanck smear was prepared from scrapings of the aforementioned lesions by the attending physician, and multinucleated epithelial giant cells were observed microscopically. According to the clinical presentation and histologic finding, which viral infection should be suspected in this case?
A 16-year-old male presents to his pediatrician with a sore throat. He reports a severely painful throat preceded by several days of malaise and fatigue. He has a history of seasonal allergies and asthma. The patient is a high school student and is on the school wrestling team. He takes cetirizine and albuterol. His temperature is 100.9°F (38.3°C), blood pressure is 100/70 mmHg, pulse is 100/min, and respirations are 20/min. Physical examination reveals splenomegaly and posterior cervical lymphadenopathy. Laboratory analysis reveals the following: Serum: Na+: 145 mEq/L K+: 4.0 mEq/L Cl-: 100 mEq/L HCO3-: 24 mEq/L BUN: 12 mg/dL Ca2+: 10.2 mg/dL Mg2+: 2.0 mEq/L Creatinine: 1.0 mg/dL Glucose: 77 mg/dL Hemoglobin: 17 g/dL Hematocrit: 47% Mean corpuscular volume: 90 µm3 Reticulocyte count: 1.0% Platelet count: 250,000/mm3 Leukocyte count: 13,000/mm3 Neutrophil: 45% Lymphocyte: 42% Monocyte: 12% Eosinophil: 1% Basophil: 0% Which of the following cell surface markers is bound by the pathogen responsible for this patient’s condition?
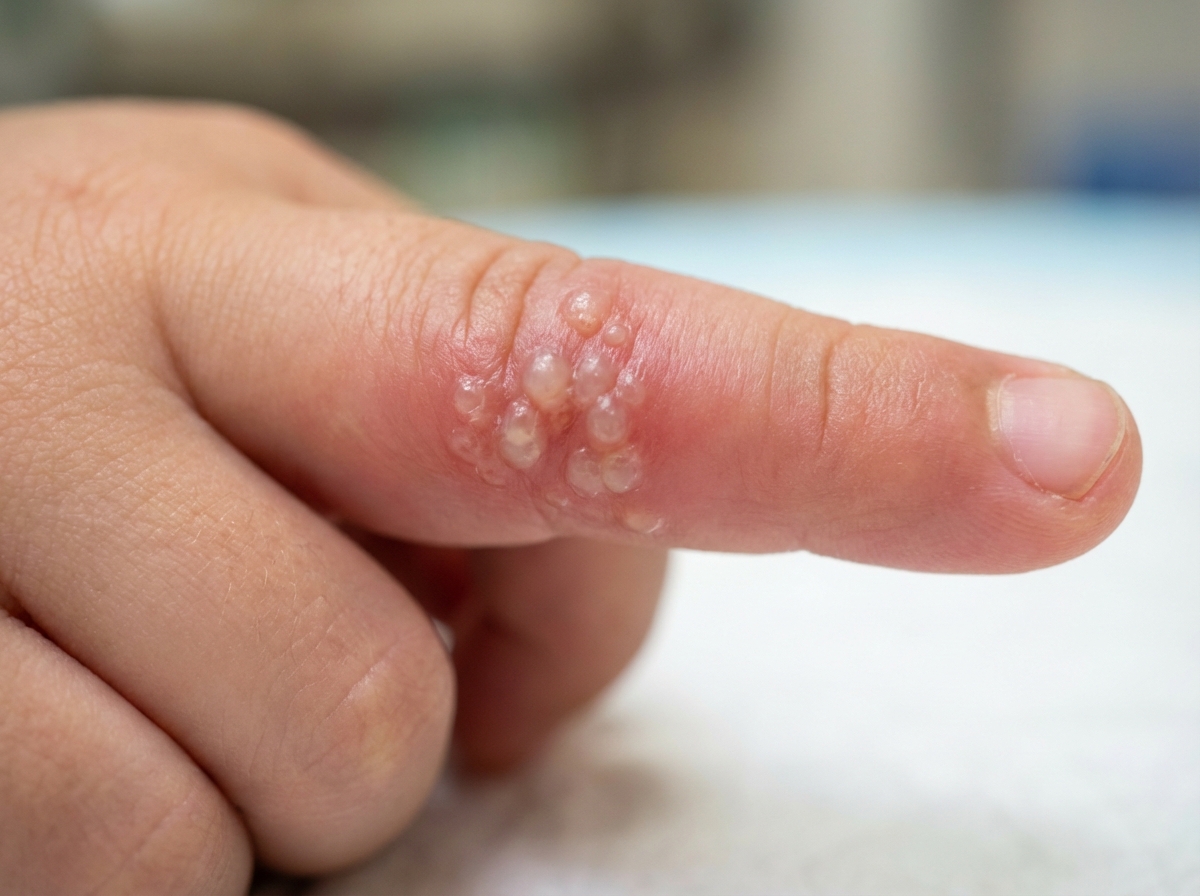
A 15-month-old girl is brought to the physician because of a 2-day history of low-grade fever and a painful lesion on her right index finger. She was born at term and has been healthy except for a rash on her upper lip 2 weeks ago, which resolved without treatment. She lives at home with her parents, her 5-year-old brother, and two cats. Her temperature is 38.5°C (101.3°F), pulse is 110/min, respirations are 30/min, and blood pressure is 100/70 mm Hg. A photograph of the right index finger is shown. Physical examination shows tender left epitrochlear lymphadenopathy. Which of the following is the most likely causal organism?
A previously healthy 16-year-old girl comes to the physician because of fever, fatigue, and a sore throat for 8 days. She also has a diffuse rash that started yesterday. Three days ago, she took amoxicillin that she had at home. She is sexually active with two male partners and uses condoms inconsistently. Her temperature is 38.4°C (101.1°F), pulse 99/min, blood pressure 106/70 mm Hg. Examination shows a morbilliform rash over her trunk and extremities. Oropharyngeal examination shows tonsillar enlargement and erythema with exudates. Tender cervical and inguinal lymphadenopathy are present. Abdominal examination shows mild splenomegaly. A peripheral blood smear shows lymphocytosis with > 10% atypical lymphocytes. Which of the following is most likely to be positive in this patient?
A 36-year-old woman presents to the emergency department with a 2-day history of conjunctivitis, sensitivity to bright light, and decreased visual acuity. She denies a history of ocular trauma. She wears contact lenses and thought that the contact lenses may be the cause of the symptoms, although she has always used proper hygiene. Fluorescein staining showed a corneal dendritic branching ulcer with terminal bulbs that stained with rose bengal. Giemsa staining revealed multinucleated giant cells. What is the most likely causative agent?
A 35-year-old male nurse presents to the emergency room complaining of fever and malaise. He recently returned from a medical trip to Liberia to help with a deadly outbreak of a highly infectious disease. He reports severe generalized muscle pain, malaise, fatigue, and a sore throat. He has recently developed some difficulty breathing and a nonproductive cough. His past medical history is notable for asthma. He drinks alcohol socially and does not smoke. His temperature is 102.1°F (38.9°C), blood pressure is 115/70 mmHg, pulse is 115/min, and respirations are 24/min. On examination, a generalized maculopapular rash and bilateral conjunctival injection are noted. Laboratory testing reveals the presence of negative sense, singled-stranded linear genetic material with filaments of varying lengths. The pathogen responsible for this patient’s symptoms is most similar to which of the following?
An investigator is studying the structural characteristics of pathogenic viruses. Cell cultures infected by different viruses are observed under a scanning electron microscope. One of the cell samples is infected by a virus that has an envelope composed of nuclear membrane molecules. The most likely virus that has infected this cell sample can cause which of the following conditions?
A 25-year-old G1P0 gives birth to a male infant at 33 weeks’ gestation. The mother immigrated from Sudan one month prior to giving birth. She had no prenatal care and took no prenatal vitamins. She does not speak English and is unable to provide a medical history. The child’s temperature is 101.0°F (38.3°C), blood pressure is 90/50 mmHg, pulse is 140/min, and respirations are 30/min. Physical examination reveals flexed upper and lower extremities, minimal response to stimulation, and slow and irregular respirations. A murmur is best heard over the left second intercostal space. The child’s lenses appear pearly white. Which of the following classes of pathogens is most likely responsible for this patient’s condition?
A 15-year-old high school rugby player presents to your clinic with a sore throat. He reports that he started feeling fatigued along with body aches about a week ago. His vitals and physical are normal except for an exudative pharynx and an enlarged spleen. Monospot test comes back positive and the student is told not to participate in contact sports for a month. What is the most likely causative agent and which immune cell does it affect?
A 14-year-old boy is brought to the physician with fever, malaise, and bilateral facial pain and swelling that began 2 days ago. He has no history of serious illness and takes no medications. He was born in India, and his mother received no prenatal care. She is unsure of his childhood vaccination history. He returned from a trip to India 3 weeks ago, where he was visiting his family. His temperature is 38.2°C (100.8°F). There is erythema, edema, and tenderness of the right and left parotid glands. The remainder of the examination shows no abnormalities. Laboratory studies show: Leukocyte count 13,000/mm3 Hemoglobin 13.0 g/dL Hematocrit 38% Platelet count 180,000/mm3 This patient is at greatest risk for which of the following complications?
Practice by Chapter
Viral structure and classification
Practice Questions
Viral replication cycles
Practice Questions
Herpesviruses (HSV, VZV, CMV, EBV, HHV-6/7/8)
Practice Questions
Respiratory viruses (influenza, RSV, parainfluenza)
Practice Questions
Enteroviruses and parechoviruses
Practice Questions
Gastroenteritis viruses (norovirus, rotavirus)
Practice Questions
Arboviruses (dengue, Zika, chikungunya)
Practice Questions
Viral hemorrhagic fevers
Practice Questions
Rabies virus
Practice Questions
Poxviruses
Practice Questions
Antiviral agents and mechanisms
Practice Questions
Viral vaccines
Practice Questions
Viral diagnostics
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
