Factorial designs — MCQs

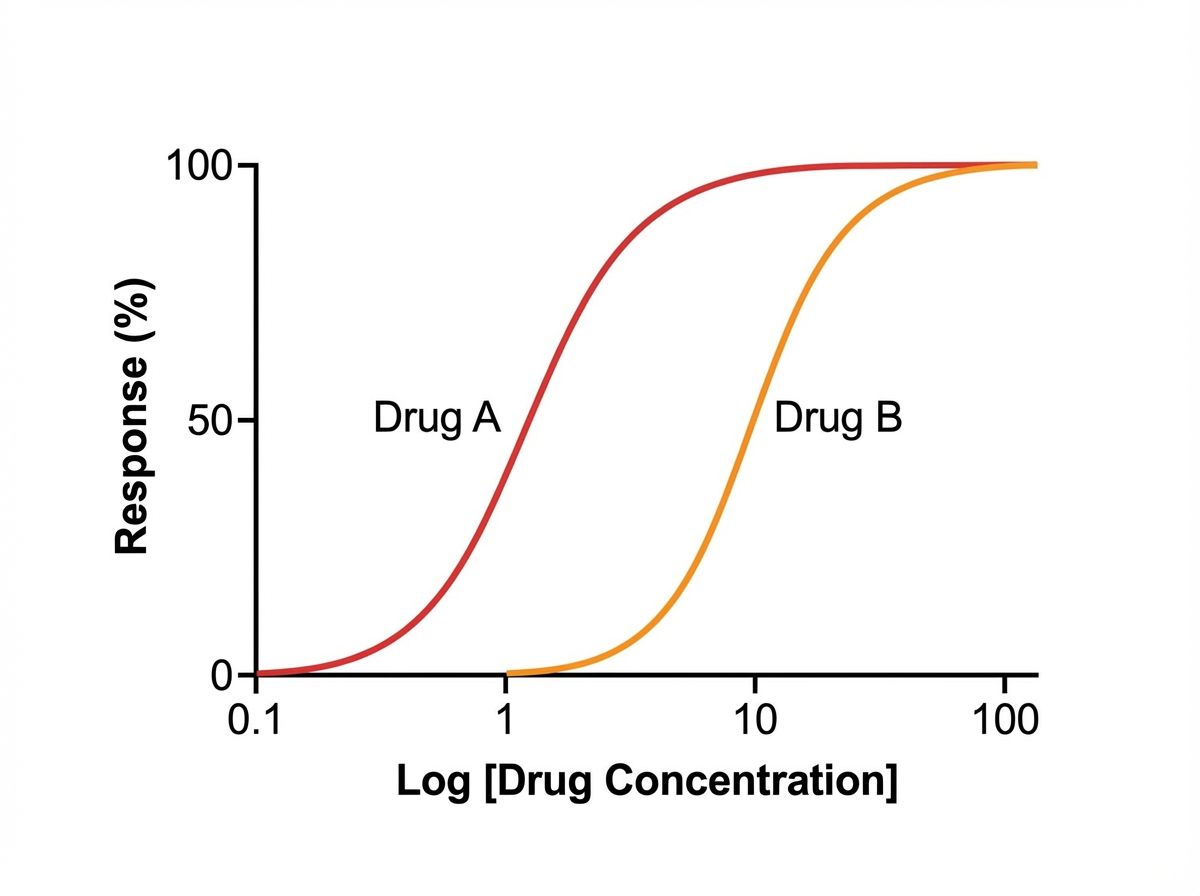
A student is reviewing the various effects that can be plotted on a dose-response curve. He has observed that certain drugs can work as an agonist and an antagonist at a particular site. He has plotted a particular graph (as shown below) and is checking for other responses that can be measured on the same graph. He learned that drug B is less potent than drug A. Drug B also reduces the potency of drug A when combined in the same solution; however, if additional drug A is added to the solution, the maximal efficacy (Emax) of drug A increases. He wishes to plot another curve for drug C. He learns that drug C works on the same molecules as drugs A and B, but drug C reduces the maximal efficacy (Emax) of drug A significantly when combined with drug A. Which of the following best describes drug C?
Researchers are studying the effects of a new medication for the treatment of type 2 diabetes. A randomized group of 100 subjects is given the new medication 1st for 2 months, followed by a washout period of 2 weeks, and then administration of the gold standard medication for 2 months. Another randomized group of 100 subjects is given the gold standard medication 1st for 2 months, followed by a washout period of 2 weeks, and then administration of the new medication for 2 months. What is the main disadvantage of this study design?
A researcher is trying to determine whether a newly discovered substance X can be useful in promoting wound healing after surgery. She conducts this study by enrolling the next 100 patients that will be undergoing this surgery and separating them into 2 groups. She decides which patient will be in which group by using a random number generator. Subsequently, she prepares 1 set of syringes with the novel substance X and 1 set of syringes with a saline control. Both of these sets of syringes are unlabeled and the substances inside cannot be distinguished. She gives the surgeon performing the surgery 1 of the syringes and does not inform him nor the patient which syringe was used. After the study is complete, she analyzes all the data that was collected and performs statistical analysis. This study most likely provides which level of evidence for use of substance X?
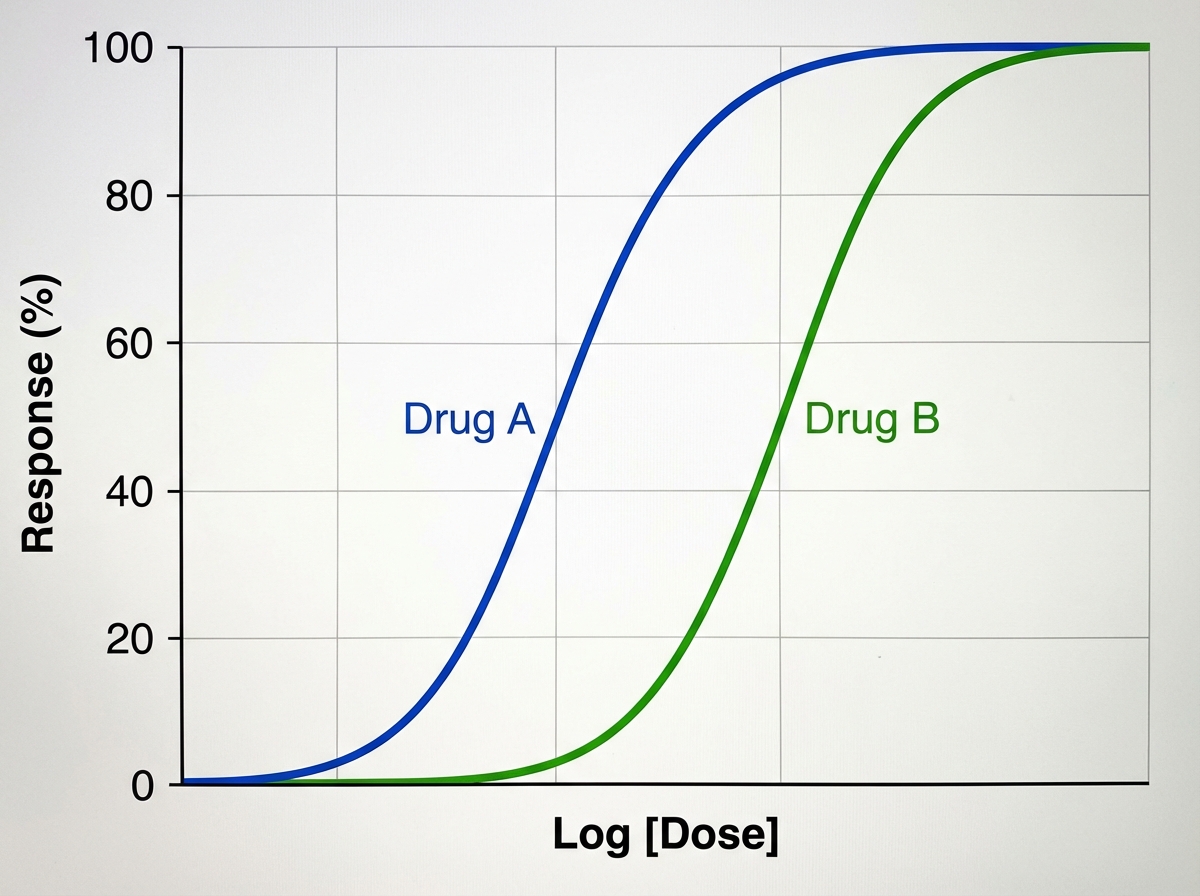
A medical student is reviewing dose-response curves of various experimental drugs. She is specifically interested in the different factors that cause the curve to shift in different directions. From her study, she plots the following graph (see image). She marks the blue curve for drug A, which acts optimally on a receptor. After drawing the second (green) curve for drug B, she discovers that more of drug B is required to produce the same response as drug A, although drug B can still achieve the same maximum effect. Which of the following terms best describes the activity of drug B in comparison to drug A?
A pharmaceutical company conducts a randomized clinical trial in an attempt to show that their new anticoagulant drug prevents more thrombotic events following total knee arthroplasty than the current standard of care. However, a significant number of patients are lost to follow-up or fail to complete treatment according to the study arm to which they were assigned. Several patients in the novel drug arm are also switched at a later time to a novel anticoagulant or warfarin per their primary care physician. All patients enrolled in the study are subsequently analyzed based on the initial group they were assigned to and there is a significant improvement in outcome of the new drug. What analysis most appropriately describes this trial?
A 28-year-old male presents to his primary care physician with complaints of intermittent abdominal pain and alternating bouts of constipation and diarrhea. His medical chart is not significant for any past medical problems or prior surgeries. He is not prescribed any current medications. Which of the following questions would be the most useful next question in eliciting further history from this patient?
You are interested in studying the etiology of heart failure reduced ejection fraction (HFrEF) and attempt to construct an appropriate design study. Specifically, you wish to look for potential causality between dietary glucose consumption and HFrEF. Which of the following study designs would allow you to assess for and determine this causality?
A clinical trial is conducted to determine the efficacy of ginkgo biloba in the treatment of Parkinson disease. A sample of patients with Parkinson disease is divided into two groups. Participants in the first group are treated with ginkgo biloba, and participants in the other group receive a placebo. A change in the Movement Disorder Society-Unified Parkinson Disease Rating Scale (MDS-UPDRS) score is used as the primary endpoint for the study. The investigators, participants, and data analysts were meant to be blinded throughout the trial. However, while the trial is being conducted, the patients' demographics and their allocated treatment groups are mistakenly disclosed to the investigators, but not to the participants or the data analysts, because of a technical flaw. The study concludes that there is a significant decrease in MDS-UPDRS scores in patients treated with ginkgo biloba. Which of the following is most likely to have affected the validity of this study?
A study seeks to investigate the therapeutic efficacy of treating asymptomatic subclinical hypothyroidism in preventing symptoms of hypothyroidism. The investigators found 300 asymptomatic patients with subclinical hypothyroidism, defined as serum thyroid-stimulating hormone (TSH) of 5 to 10 μU/mL with normal serum thyroxine (T4) levels. The patients were randomized to either thyroxine 75 μg daily or placebo. Both investigators and study subjects were blinded. Baseline patient characteristics were distributed similarly in the treatment and control group (p > 0.05). Participants' serum T4 and TSH levels and subjective quality of life were evaluated at a 3-week follow-up. No difference was found between the treatment and placebo groups. Which of the following is the most likely explanation for the results of this study?
In the study, all participants who were enrolled and randomly assigned to treatment with pulmharkimab were analyzed in the pulmharkimab group regardless of medication nonadherence or refusal of allocated treatment. A medical student reading the abstract is confused about why some participants assigned to pulmharkimab who did not adhere to the regimen were still analyzed as part of the pulmharkimab group. Which of the following best reflects the purpose of such an analysis strategy?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
