Lipid metabolism — MCQs

On this page
Colipase is an enzyme found in secretions from which of the following? (Recent NEET Pattern 2018)
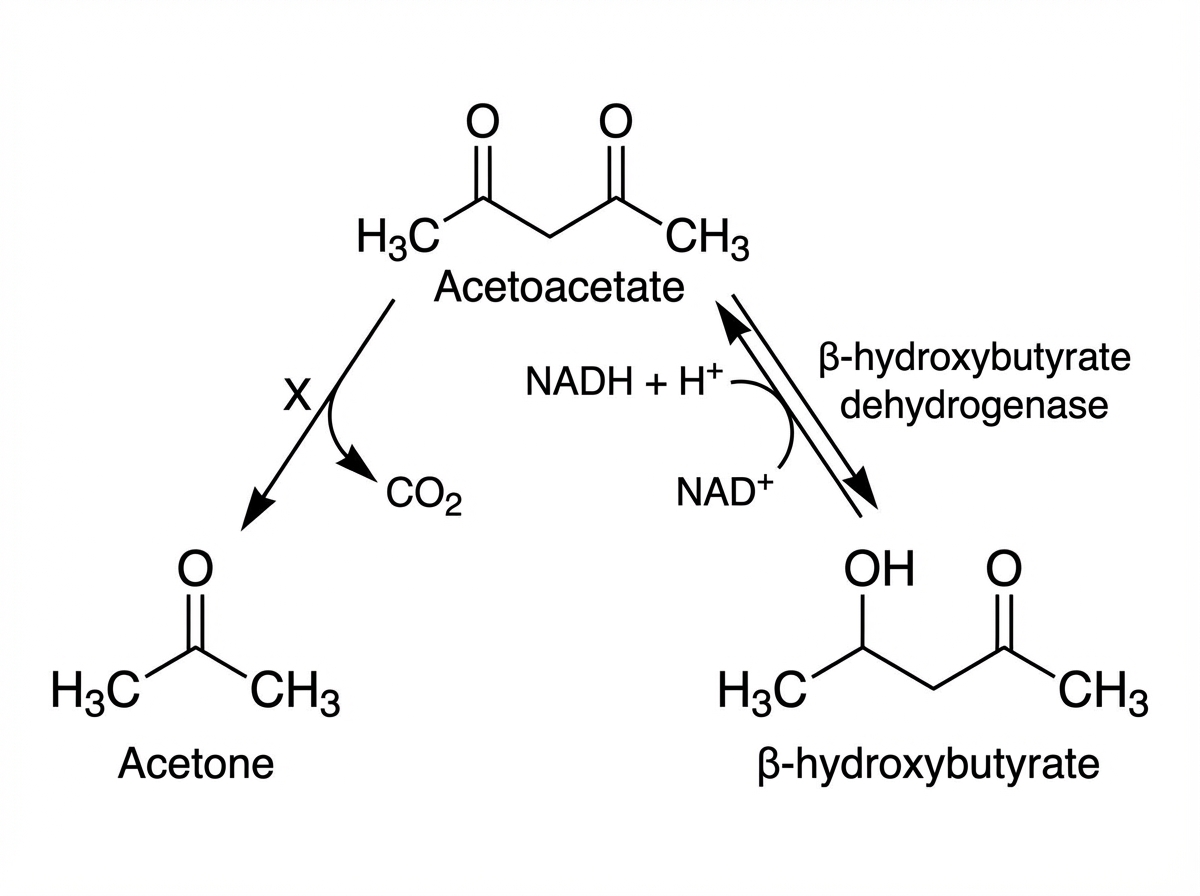
Name the mechanism shown as $X$ below.
Which is the location of HMG CoA synthase in cholesterol metabolism?
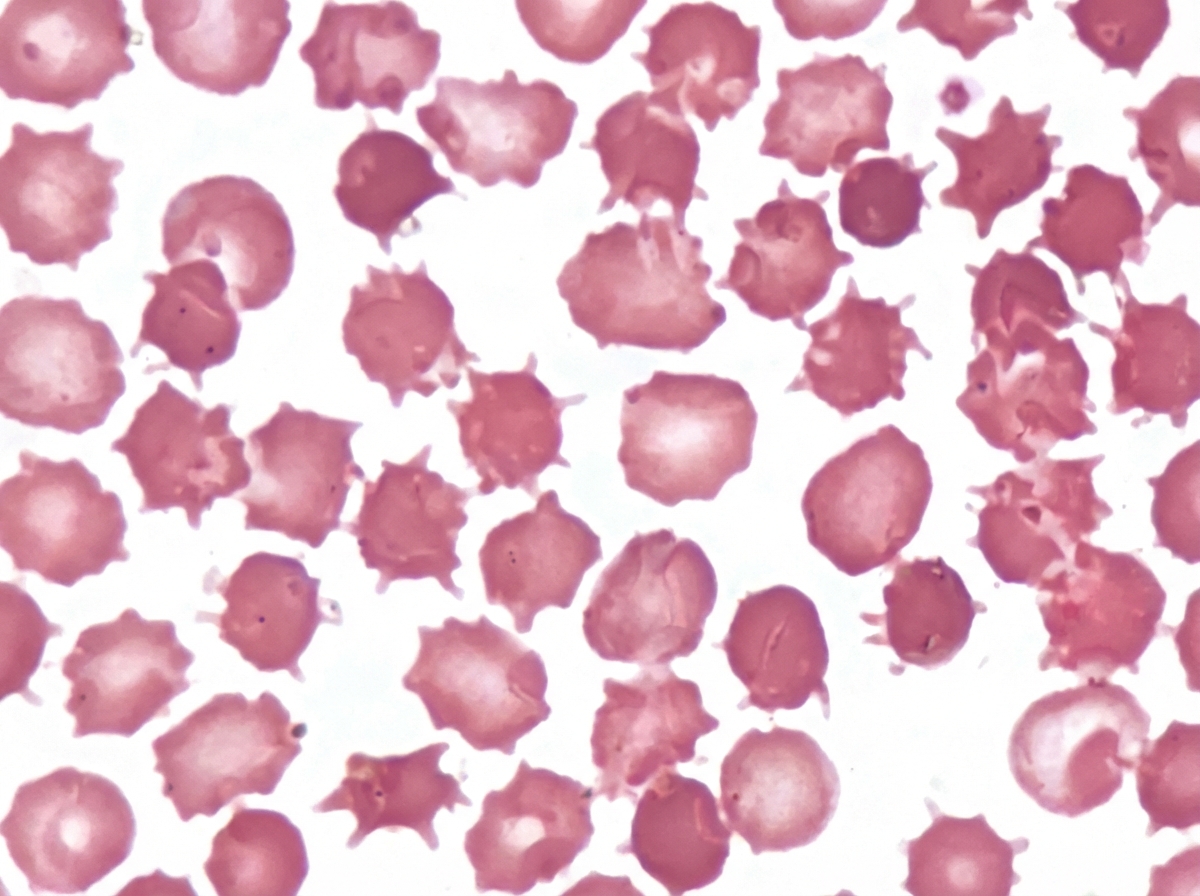
An adult male presented with a protruding abdomen, diarrhea, visual symptoms, and neurological manifestations. His LDL is low. Based on the peripheral smear finding shown in the image, what is the likely diagnosis?
A patient came to the hospital with severe abdominal pain, and lipase levels were elevated. On imaging, a stone is found in the common bile duct (CBD). Which enzyme is most likely elevated in this condition?
Practice by Chapter
Fatty acid oxidation (beta-oxidation)
Practice Questions
Fatty acid synthesis
Practice Questions
Ketone body metabolism
Practice Questions
Cholesterol synthesis and regulation
Practice Questions
Lipoprotein metabolism
Practice Questions
Phospholipid metabolism
Practice Questions
Eicosanoid synthesis and function
Practice Questions
Steroid hormone synthesis
Practice Questions
Adipose tissue metabolism
Practice Questions
Brown vs. white adipose tissue
Practice Questions
Disorders of lipid metabolism
Practice Questions
Integration with carbohydrate metabolism
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
