Glycolysis — MCQs

On this page
A 16-year-old teenager is brought to the emergency department after having slipped on ice while walking to school. She hit her head on the side of the pavement and retained consciousness. She was brought to the closest ER within an hour of the incident. The ER physician sends her immediately to get a CT scan and also orders routine blood work. The physician understands that in cases of stress, such as in this patient, the concentration of certain hormones will be increased, while others will be decreased. Considering allosteric regulation by hormones, which of the following enzymes will most likely be inhibited in this patient?
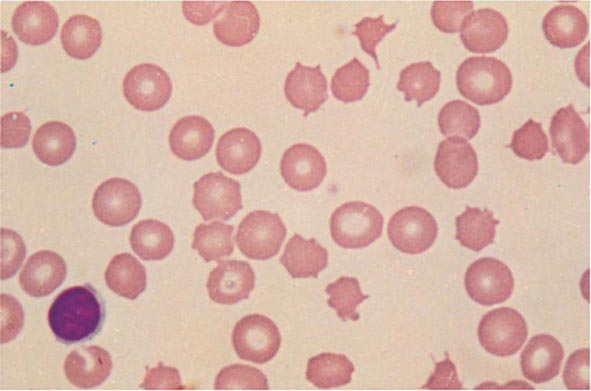
A mother brings her newborn baby to the pediatrician after noting that his skin looks yellow. The patient's lactate dehydrogenase is elevated and haptoglobin is decreased. A smear of the child's blood is shown below. The patient is ultimately found to have decreased ability to process phosphoenolpyruvate to pyruvate. Which of the following metabolic changes is most likely to occur in this patient?
Researchers are investigating a new mouse model of glycogen regulation. They add hepatocyte enzyme extracts to radiolabeled glucose to investigate glycogen synthesis, in particular two enzymes. They notice that the first enzyme adds a radiolabeled glucose to the end of a long strand of radiolabeled glucose. The second enzyme then appears to rearrange the glycogen structure such that there appears to be shorter strands that are linked. Which of the following pairs of enzymes in humans is most similar to the enzymes being investigated by the scientists?
Maturity Onset Diabetes of the Young (MODY) type 2 is a consequence of a defective pancreatic enzyme, which normally acts as a glucose sensor, resulting in a mild hyperglycemia. The hyperglycemia is especially exacerbated during pregnancy. Which of the following pathways is controlled by this enzyme?
To maintain blood glucose levels even after glycogen stores have been depleted, the body, mainly the liver, is able to synthesize glucose in a process called gluconeogenesis. Which of the following reactions of gluconeogenesis requires an enzyme different from glycolysis?
Practice by Chapter
Overview and regulation of glycolysis
Practice Questions
Preparatory phase reactions
Practice Questions
Pay-off phase reactions
Practice Questions
Rate-limiting enzymes and control points
Practice Questions
Energy yield and ATP production
Practice Questions
Fates of pyruvate
Practice Questions
Substrate-level phosphorylation
Practice Questions
Feeder pathways to glycolysis
Practice Questions
Glycolysis in different tissues
Practice Questions
Disorders of glycolytic enzymes
Practice Questions
Alternative glycolytic pathways
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
