Transplant Surgery — MCQs

On this page
Which type of Maastricht classification is a brought dead patient?
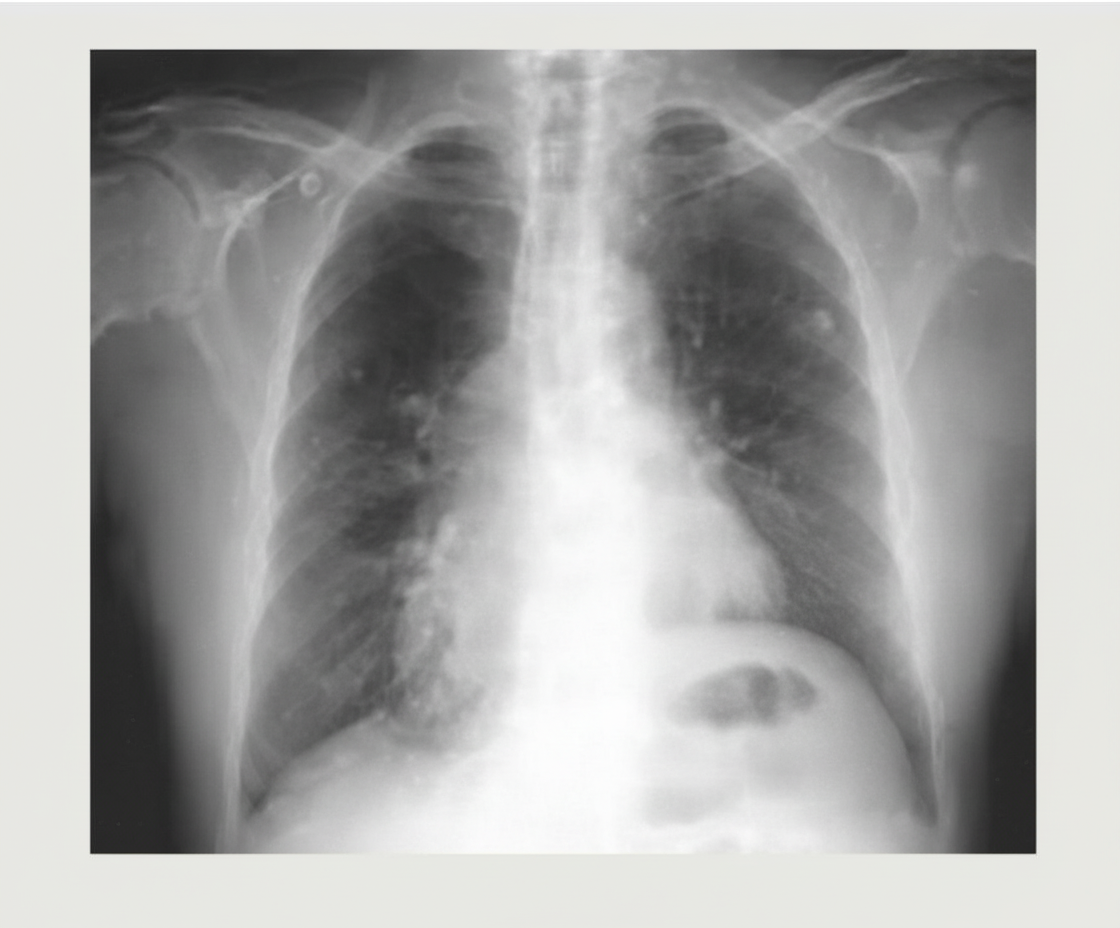
A 32-year-old male who underwent a lung transplant 1 year ago, with a history of acute rejection requiring increased immunosuppression, now presents with low-grade fever, generalized fatigue, and weight loss. Chest x-rays are obtained. What is the most likely diagnosis?
Who performed the first autologous renal transplantation?
Recovery of renal functions after renal transplant usually takes how long?
Hypothermia (0deg to 4deg C) is a critical component of successful organ cold storage because:
Which disease does not recur in the kidney after renal transplant?
What is considered a contraindication for liver transplant?
Highest chance of success in renal transplant is seen when the donor is:
Which of the following organs obtained from a cadaver is not used for transplant?
Which of the following is a criterion for a pancreas donor?
Practice by Chapter
Immunology of Transplantation
Practice Questions
Immunosuppression
Practice Questions
Organ Procurement
Practice Questions
Kidney Transplantation
Practice Questions
Liver Transplantation
Practice Questions
Pancreas Transplantation
Practice Questions
Heart Transplantation
Practice Questions
Lung Transplantation
Practice Questions
Small Bowel Transplantation
Practice Questions
Complications of Transplantation
Practice Questions
Transplantation in Special Populations
Practice Questions
Ethical Issues in Transplantation
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
