Pancreatic Surgery — MCQs

On this page
Which of the following is NOT a cause of acute pancreatitis?
Pancreatic pseudocyst most commonly occurs after which condition?
What is the commonest cyst to arise in the pancreas after an attack of acute pancreatitis or pancreatic trauma?
Insulinoma is most commonly located in which part of the pancreas?
True about Insulinoma is:
A chronic alcoholic presented with repeated episodes of non-bilious vomiting after meals. Based on CECT findings, what is the diagnosis?
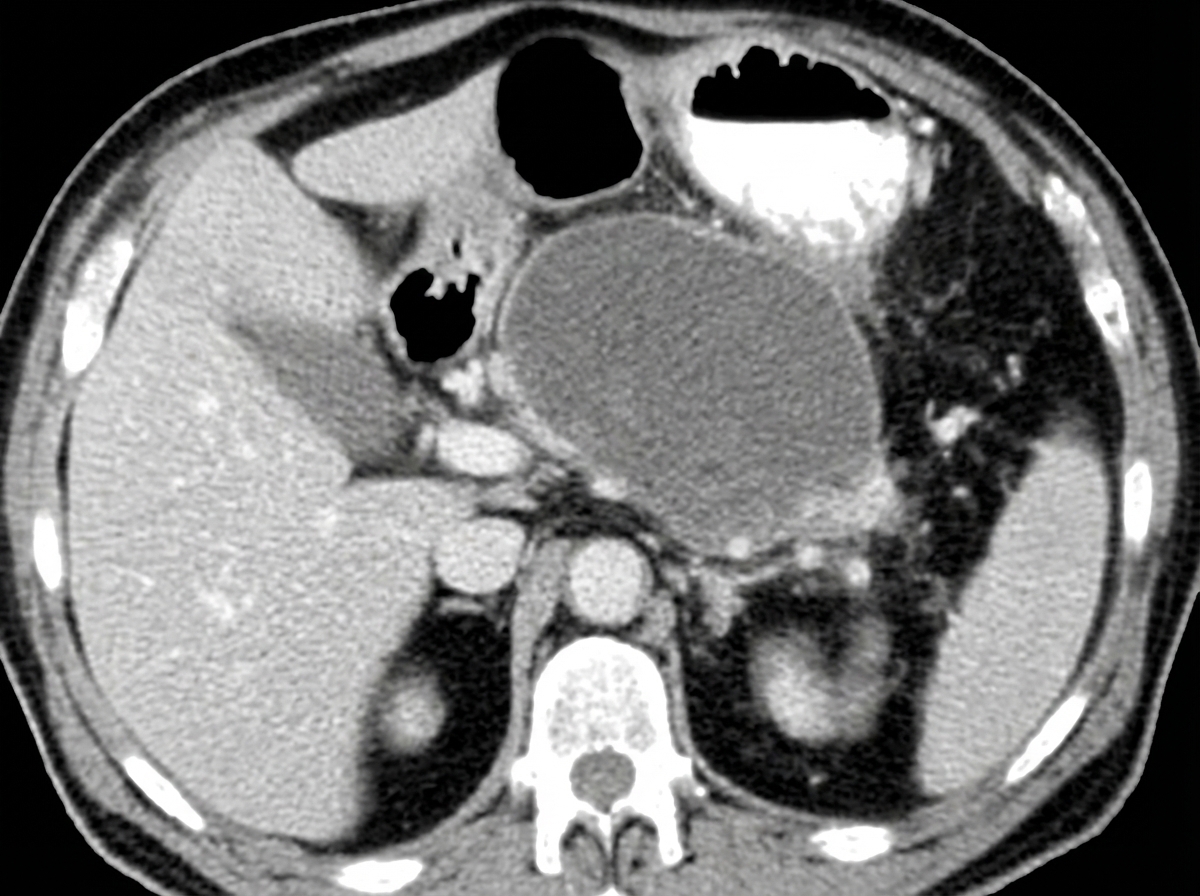
Which branch of the portal vein is involved in chronic pancreatitis?
Which of the following is NOT a cause of acute pancreatitis?
A 25-year-old female presents with episodes of bizarre behavior, memory lapse, and unconsciousness. She also demonstrated previous episodes of extreme hunger, sweating, and tachycardia. During one of these episodes, her blood sugar was tested and was found to be 40 mg/dL. Which of the following would most appropriately indicate a diagnosis of insulinoma?
Which of the following statements is NOT true regarding a pseudocyst of the pancreas?
Practice by Chapter
Pancreatic Anatomy and Physiology
Practice Questions
Acute Pancreatitis
Practice Questions
Chronic Pancreatitis
Practice Questions
Pancreatic Pseudocysts
Practice Questions
Pancreatic Adenocarcinoma
Practice Questions
Cystic Neoplasms of Pancreas
Practice Questions
Neuroendocrine Tumors of Pancreas
Practice Questions
Pancreatic Trauma
Practice Questions
Pancreatectomy Techniques
Practice Questions
Whipple Procedure
Practice Questions
Pancreatic Anastomosis
Practice Questions
Complications of Pancreatic Surgery
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
