Radiation-Induced DNA Damage — MCQs

10 questions
Read Study NotesQ1
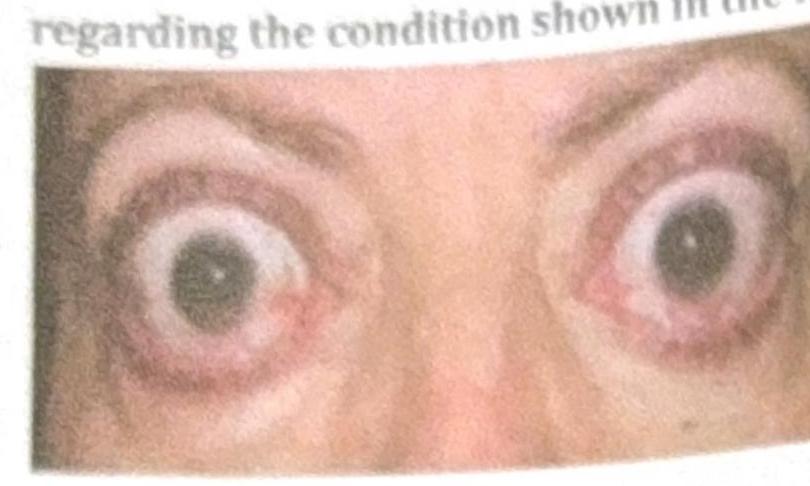
Which of the following statements about thyroid eye disease is false?
Q2
Radiation mediates its effect by
Q3
Cell most sensitive to radiation –
Q4
Which part of DNA is most susceptible to radiation?
Q5
HNPCC has defect in which
Q6
A patient presents with a skin rash that is exaggerated on sun exposure. What is the repair mechanism involved in this condition?
Q7
Cells are most sensitive to ionizing radiation during which phase?
Q8
The component of cell most affected by radiation?
Q9
Radiation causes cell death by:
Q10
Which of the following is not a risk factor for cholangiocarcinoma?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
