Radiobiology — MCQs

On this page
What is the threshold radiation dose for the hematological syndrome?
Which of the following is a late complication of radiotherapy?
Which of the following is NOT a radioprotector?
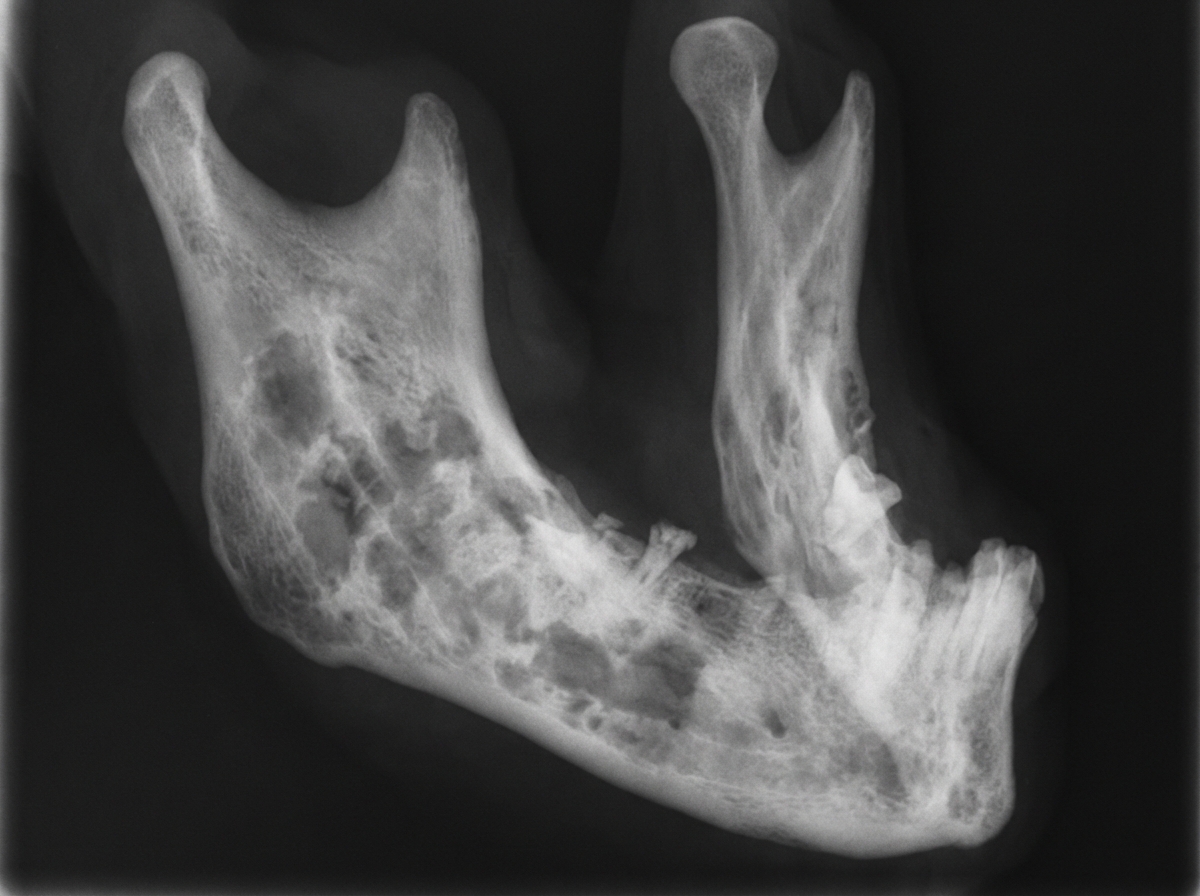
What is observed in the part of the bone which received radiotherapy?
Which of the following statements about Linear Energy Transfer (LET) is true?
Practice by Chapter
Cellular Effects of Radiation
Practice Questions
Radiation-Induced DNA Damage
Practice Questions
Cell Survival Curves
Practice Questions
Radiation Effects on Normal Tissues
Practice Questions
Acute Radiation Syndrome
Practice Questions
Late Effects of Radiation
Practice Questions
Radiotherapeutic Ratio
Practice Questions
Fractionation in Radiotherapy
Practice Questions
Oxygen Effect and Radiosensitizers
Practice Questions
Radiation Carcinogenesis
Practice Questions
Radiation in Pregnancy
Practice Questions
Biological Dosimetry
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
