Contrast-Induced Nephropathy — MCQs

All of the following are indications for hemodialysis in acute kidney injury, EXCEPT:
Which of the following is Iso-osmolar agent?
A dense persistent nephrogram may be seen in all of the following except:
Which part of the kidney is first affected by ischemia in the context of acute kidney injury?
Immediately after kidney donation what happens to the creatinine level in the donors?
Following surgery, a patient develops oliguria. You believe the patient is hypovolemic, but you seek corroborative data before increasing intravenous fluids. The best data is?
Uremic complications typically arise during which of the following phases of renal failure?
Excretory urography should be cautiously performed in
Even conventional radiological procedures are contraindicated in which neurological disease?
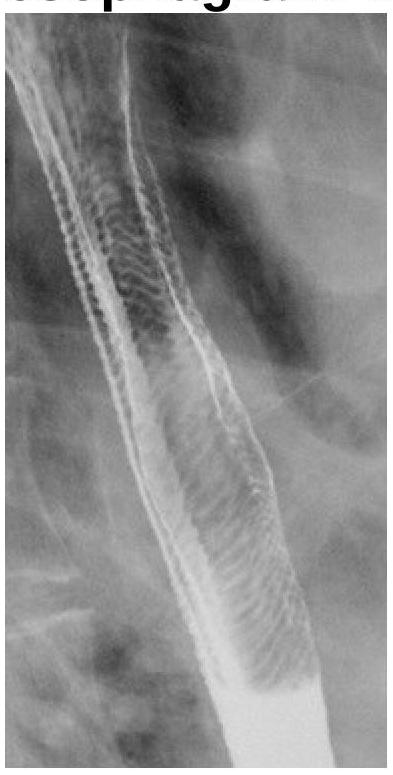
What does the following radiograph from a double contrast esophagram represent?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
