Renal Physiology — MCQs

On this page
What is the resting ureteric pressure?
Urine concentrating ability of the kidney is increased by?
What is the net filtration pressure?
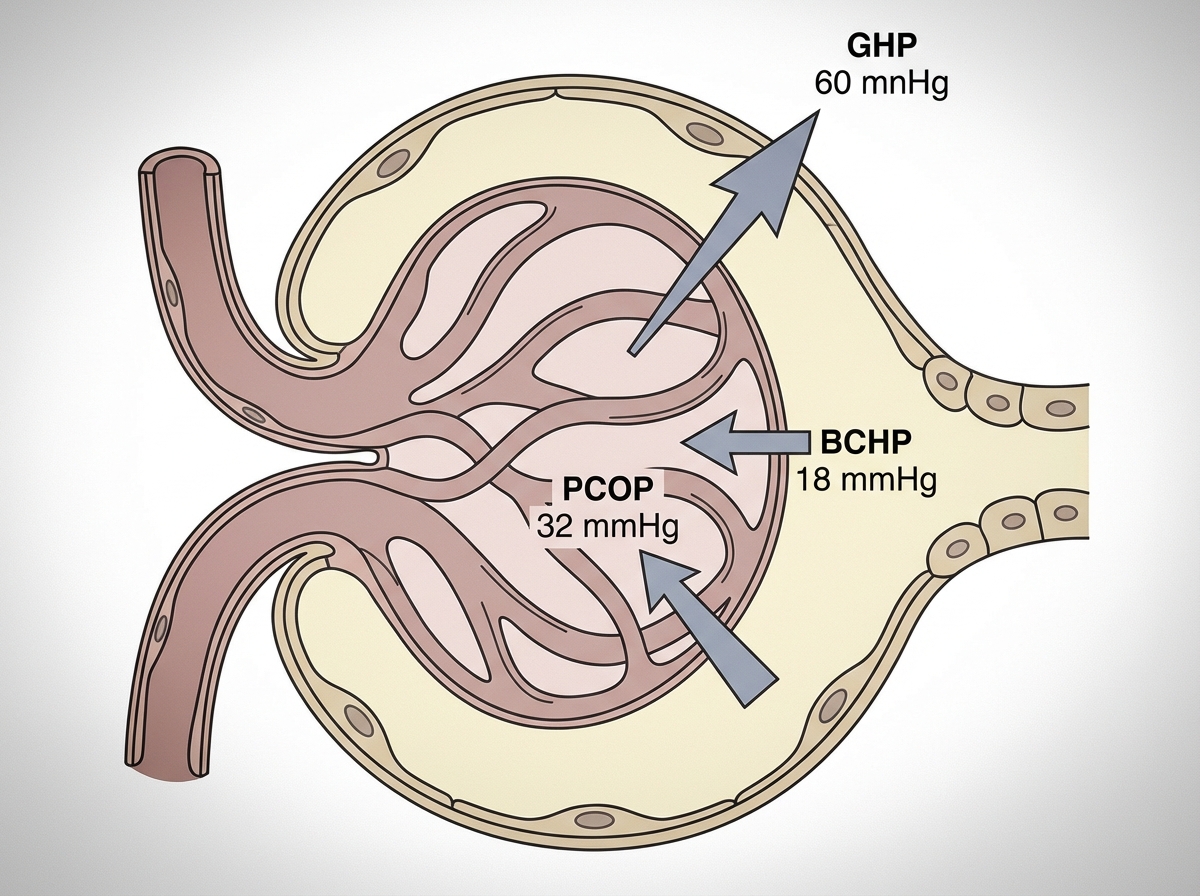
Which of the following substances does not normally pass through the glomerular filter in the kidney?
Urinary concentrating ability of the kidney is increased by?
Angiotensin-converting enzymes are?
What is the principal site of urine acidification?
Which part of the Henle loop is less permeable to water?
Why is the actual tubular maximum for the kidney in practice less than the calculated value?
In renal disease, why does albumin first appear in the urine?
Practice by Chapter
Renal Blood Flow and Glomerular Filtration
Practice Questions
Tubular Reabsorption and Secretion
Practice Questions
Concentration and Dilution of Urine
Practice Questions
Acid-Base Regulation by the Kidneys
Practice Questions
Sodium and Water Balance
Practice Questions
Potassium Regulation
Practice Questions
Calcium and Phosphate Handling
Practice Questions
Micturition Physiology
Practice Questions
Renal Function Tests
Practice Questions
Integrative Responses to Fluid Challenges
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
