Nerve and Muscle Physiology — MCQs

On this page
Arrange the following nerve fibers sequentially in the descending order of nerve impulse transmission velocity: A. C fiber B. Aa fiber C. B fiber D. Ad fiber
In Myasthenia gravis, what is the fundamental defect at the neuromuscular junction?
Where does the action potential in a spinal motor neuron generate from?
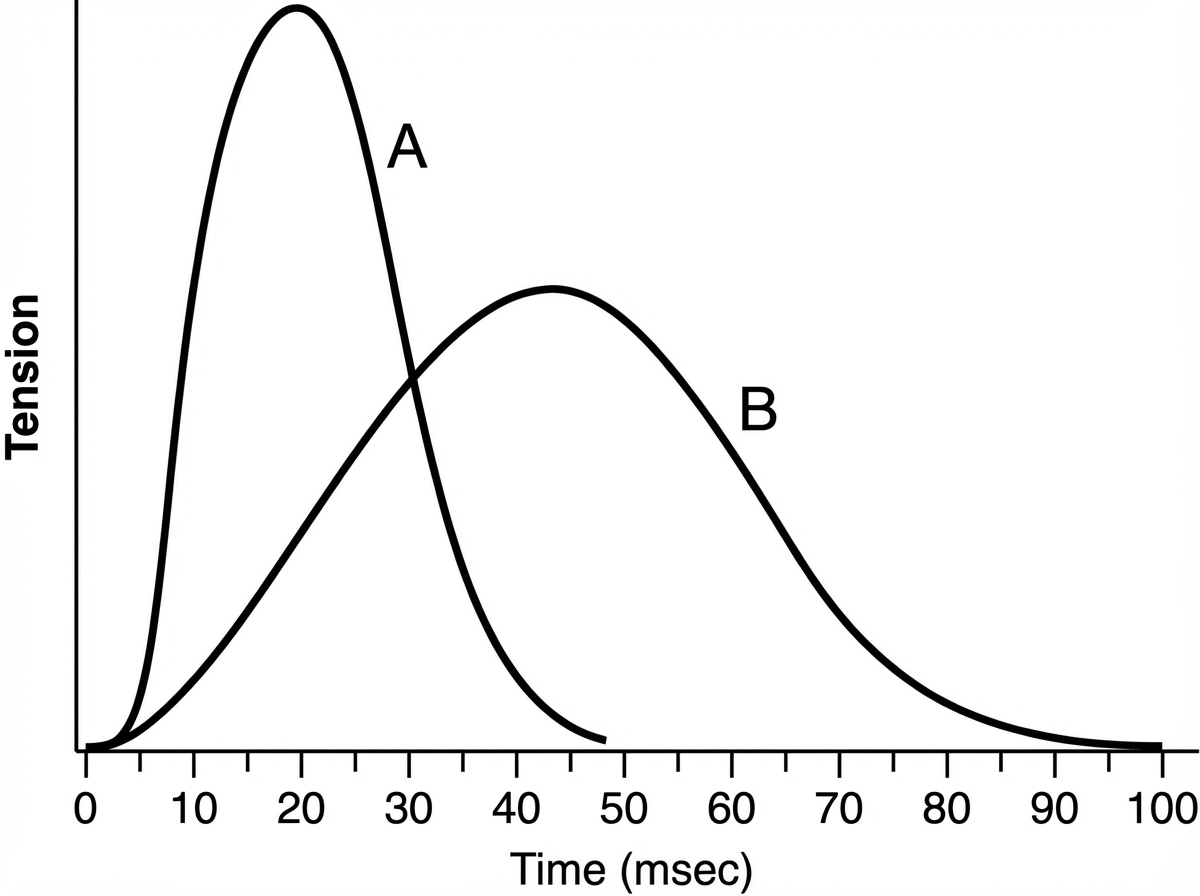
Which of the following best describes muscle B compared with muscle A?
Which of the following statements regarding intrafusal fibers is incorrect?
Post-tetanic facilitation is thought to be the result of which of the following?
Which of the following specifically blocks sodium channels?
The refractory period is minimum for which of the following nerve fibers?
Botulinum toxin blocks neuromuscular transmission by which of the following mechanisms?
Dynamic response is due to which type of muscle ending?
Practice by Chapter
Resting Membrane Potential
Practice Questions
Action Potential Generation and Propagation
Practice Questions
Neuromuscular Junction
Practice Questions
Skeletal Muscle Contraction
Practice Questions
Smooth Muscle Physiology
Practice Questions
Cardiac Muscle Properties
Practice Questions
Muscle Metabolism and Fatigue
Practice Questions
Motor Unit Function
Practice Questions
Neurotransmitters and Receptors
Practice Questions
Electrophysiological Measurements
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
