General Physiology — MCQs

On this page
What is the primary function of ubiquitin?
All of the following intercellular communications occur via extracellular chemical messengers except?
What is the term for the smooth muscle cell membrane?
Which of the following statements is true regarding basal metabolic rate?
Total body water differences between males and females are not observed at which age range?
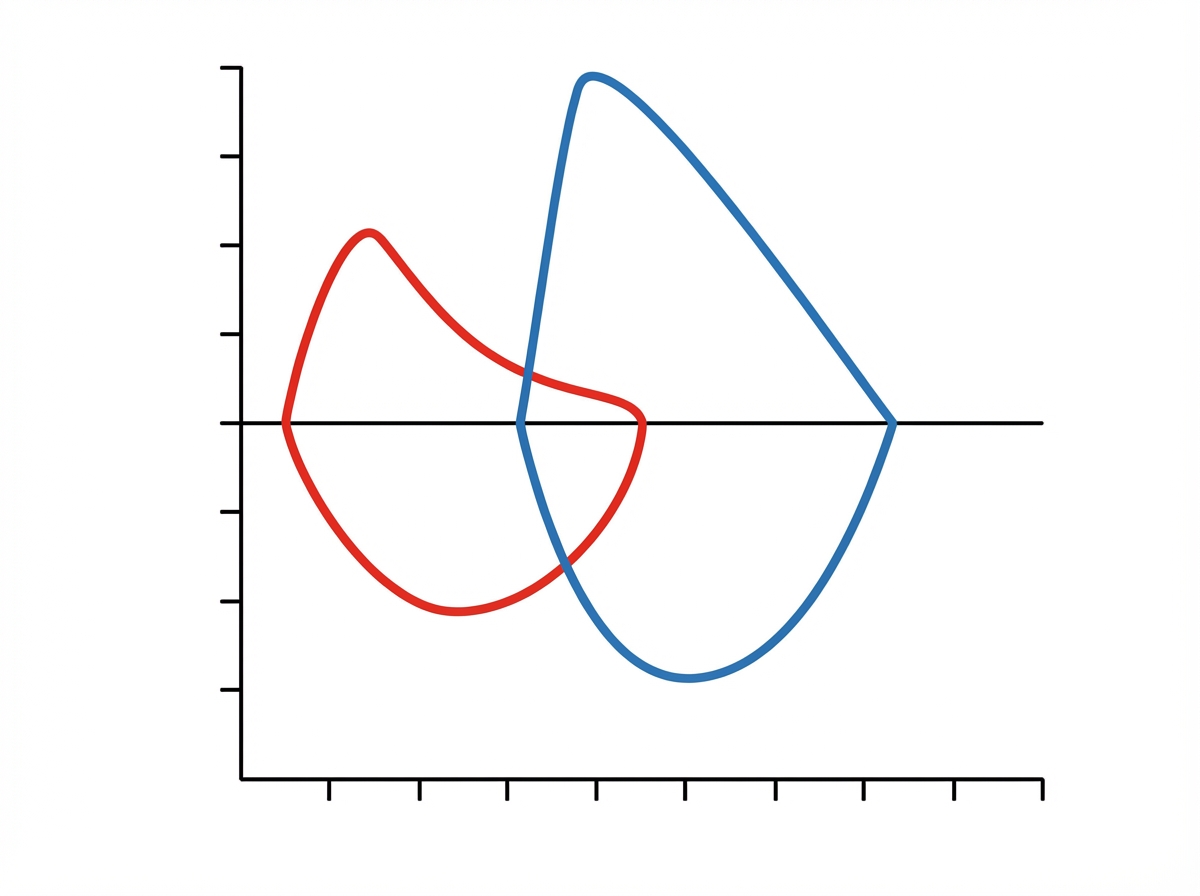
The given graph likely depicts which of the following conditions?
Within the endocrine system, specificity of communication is determined by what?
What is the reason for hyperosmolarity?
What are the functions of cytoplasmic enzymes in Red Blood Cells?
Which of the following processes requires a carrier protein?
Practice by Chapter
Cell Structure and Function
Practice Questions
Membrane Transport Mechanisms
Practice Questions
Bioelectric Phenomena
Practice Questions
Homeostasis and Feedback Mechanisms
Practice Questions
Body Fluid Compartments
Practice Questions
Signal Transduction Mechanisms
Practice Questions
Cell-to-Cell Communication
Practice Questions
Principles of Physiological Measurement
Practice Questions
Osmosis and Osmotic Pressure
Practice Questions
Physiological Adaptation Mechanisms
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
