Magnesium Metabolism — MCQs

A woman with eclampsia is started on magnesium sulfate. What is the first sign of magnesium sulfate toxicity?
Which enzyme requires zinc as a cofactor?
Hypomagnesemia due to increased excretion by the kidney is caused by all except:
Hypermagnesemia may be observed in:
Normal anion gap is___ mmol/L?
In Bartter's syndrome there is a defect in
In renal failure, what is the primary cause of metabolic acidosis?
Which of the following statements is not true?
In a child with suspected tetany, the following test is performed. Identify the sign?
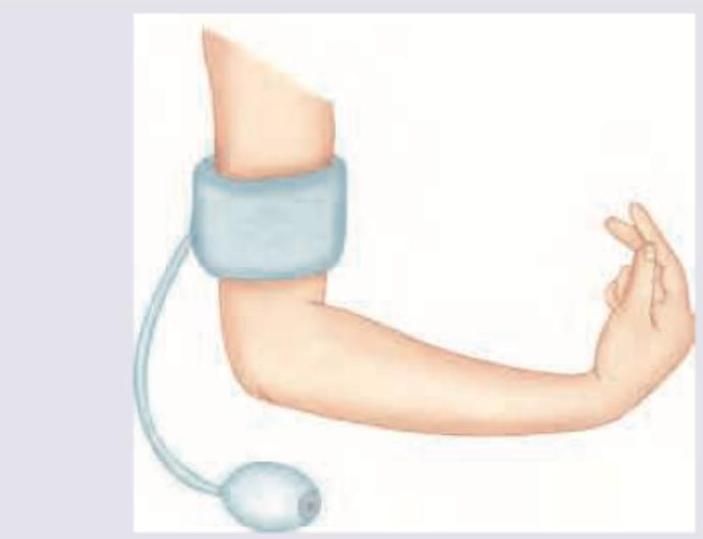
Intracellular water constitutes what percentage of total body water?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
