Blood and Immunity — MCQs

On this page
CO poisoning causes which type of hypoxia?
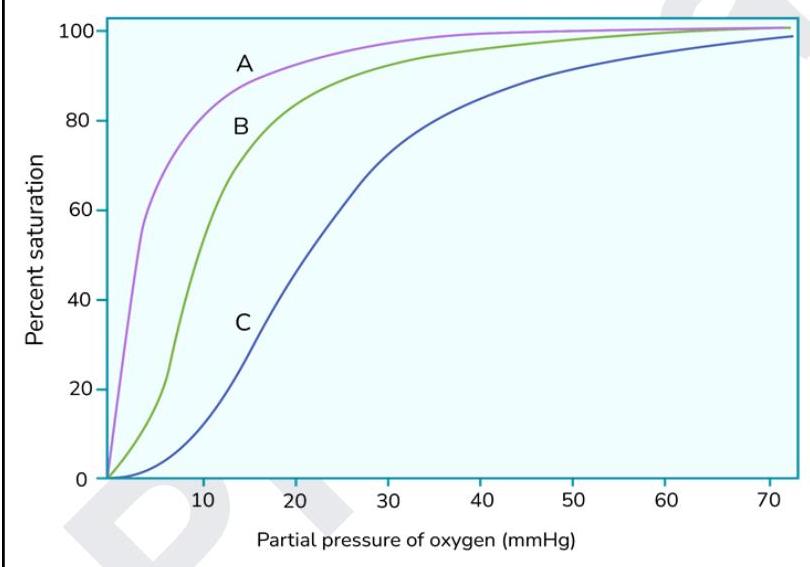
The graph below shows oxygen dissociation curves. What does the curve marked as 'A' indicate?
What is the primary stimulus for erythropoietin production?
What is needed for prothrombin to thrombin conversion?
A pregnant woman is able to transfer oxygen to her fetus because fetal hemoglobin has a greater affinity for oxygen than does adult hemoglobin. Why is the affinity of fetal hemoglobin for oxygen higher?
Decreased O2 carrying capacity and Normal PO2 is a feature of
Oxygen dependent killing is done through
Which of the following cells will increase in case of parasite infection?
Which of the following is not involved in local hemostasis?
All true about interaction of SpO2 reading and methemoglobinemia, except:
Practice by Chapter
Composition and Functions of Blood
Practice Questions
Erythrocytes and Hemoglobin
Practice Questions
Leukocytes and Immune Function
Practice Questions
Platelets and Hemostasis
Practice Questions
Blood Groups and Transfusion
Practice Questions
Coagulation and Fibrinolysis
Practice Questions
Hematopoiesis
Practice Questions
Innate Immunity
Practice Questions
Adaptive Immunity
Practice Questions
Immunological Memory and Tolerance
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
