Blood and Immunity — MCQs

On this page
A 35-year-old woman with menorrhagia presents with pallor, fatigue, and koilonychia. Laboratory findings show hemoglobin 8.2 g/dL, MCV 68 fL, serum iron 25 μg/dL, TIBC 450 μg/dL, and serum ferritin 10 ng/mL. Which of the following best explains the increased TIBC in this patient?
A macrophage engulfs different cells as shown in the image. This is known as?
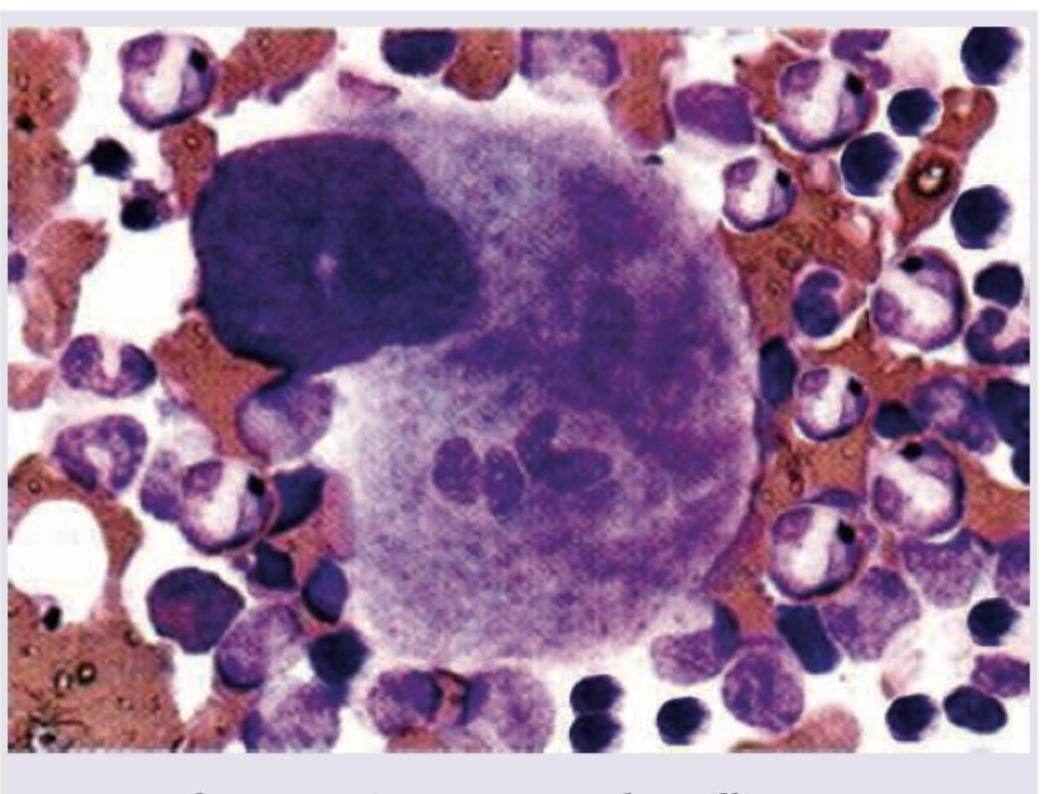
The least marked function of a human spleen is
Which of the following is X-linked dominant trait?
The genetic inheritance of Haemophilia is
Stored blood which has been preserved in a blood bank is deficient in which of the following coagulation factors?
The removal of malarial parasites from the blood is called
Which one of the following factors is NOT involved in the pathogenesis of Systemic inflammatory response syndrome (SIRS)?
The immunoglobulins that can be transported across the placenta include:
What is the ratio of T cells to B cells in a healthy adult?
Practice by Chapter
Composition and Functions of Blood
Practice Questions
Erythrocytes and Hemoglobin
Practice Questions
Leukocytes and Immune Function
Practice Questions
Platelets and Hemostasis
Practice Questions
Blood Groups and Transfusion
Practice Questions
Coagulation and Fibrinolysis
Practice Questions
Hematopoiesis
Practice Questions
Innate Immunity
Practice Questions
Adaptive Immunity
Practice Questions
Immunological Memory and Tolerance
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
