Acid-Base Balance — MCQs

On this page
What acid-base status is suggested by the given parameters?

A patient is experiencing respiratory acidosis due to brain trauma. Which of the following lab values correlates with this acid imbalance?
In the following flow diagram, Box A represents:
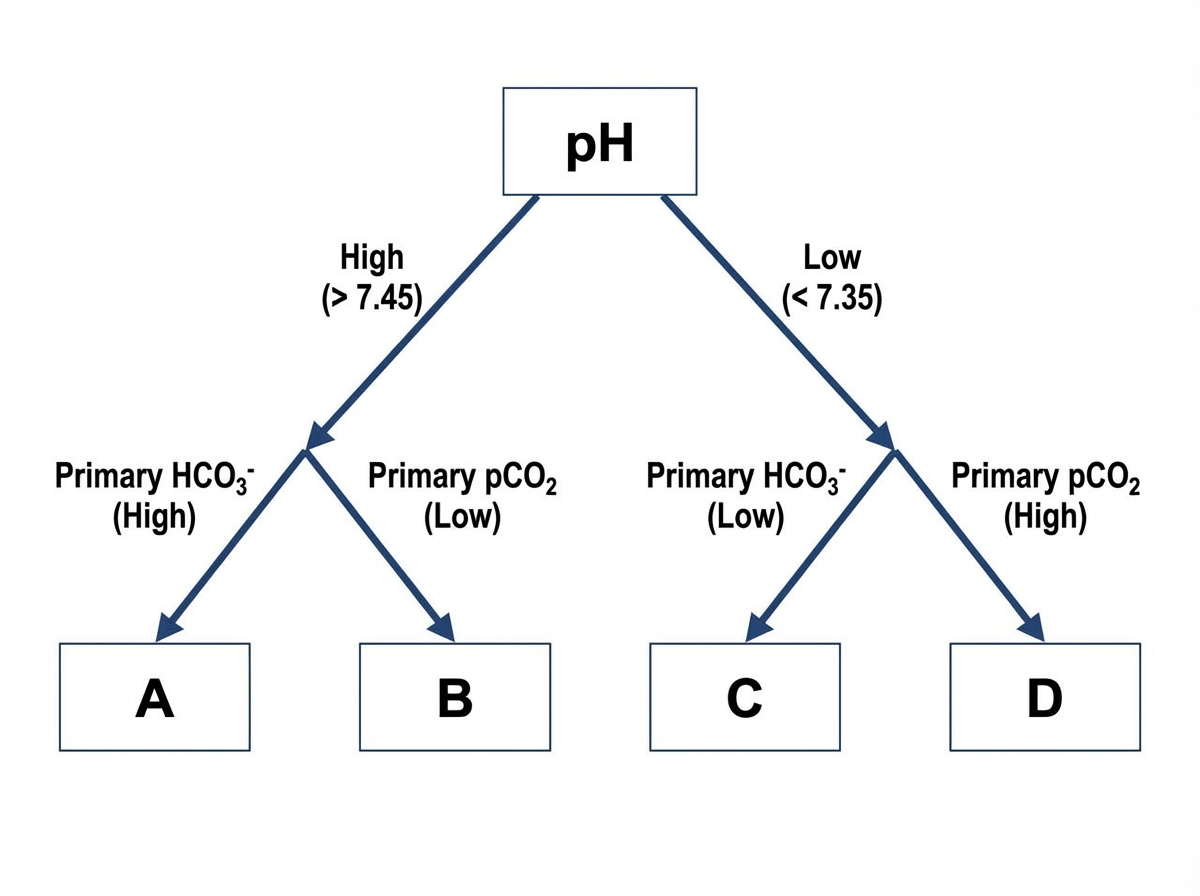
A patient's arterial blood gas analysis reveals pH 7.2, HCO3 36mmol/L, and pCO2 60mm of Hg. What is the primary acid-base abnormality?
Which of the following hormones does NOT modify growth?
Antral obstruction with vomiting is NOT characterized by which of the following?
Arterial blood gas analysis of a patient admitted to the medical emergency is as follows: pH = 7.2, HCO3 = 38 mmol/L, pCO2 = 56 mm Hg. This indicates:
Kussmaul's breathing is characteristic of which condition?
What are the ECG changes seen in hypocalcemia?
What is the commonest cause of metabolic alkalosis?
Practice by Chapter
Acid-Base Chemistry
Practice Questions
Respiratory Regulation of Acid-Base Balance
Practice Questions
Renal Regulation of Acid-Base Balance
Practice Questions
Bicarbonate Buffer System
Practice Questions
Non-Bicarbonate Buffer Systems
Practice Questions
Respiratory Acidosis and Alkalosis
Practice Questions
Metabolic Acidosis and Alkalosis
Practice Questions
Mixed Acid-Base Disorders
Practice Questions
Compensatory Mechanisms
Practice Questions
Clinical Assessment of Acid-Base Status
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
