Acid-Base Balance — MCQs

On this page
Which of the following is true about the Anion Gap?
Features seen in a patient with chronic vomiting are all EXCEPT:
Hypokalemia is frequently associated with which acid-base disorder?
A 40-year-old male presents with excessive hyperventilation. Arterial blood gas analysis reveals pH 7.5, PCO2 24 mmHg, and PO2 88 mm of Hg. What is the most likely diagnosis based on these findings?
How long does it take for metabolic compensation to occur in a child with respiratory acidosis?
Which of the following is the principal buffer in interstitial fluid?
An arterial blood gas (ABG) of a patient shows decreased pH, increased pCO2, and high bicarbonate. What is the most likely diagnosis?
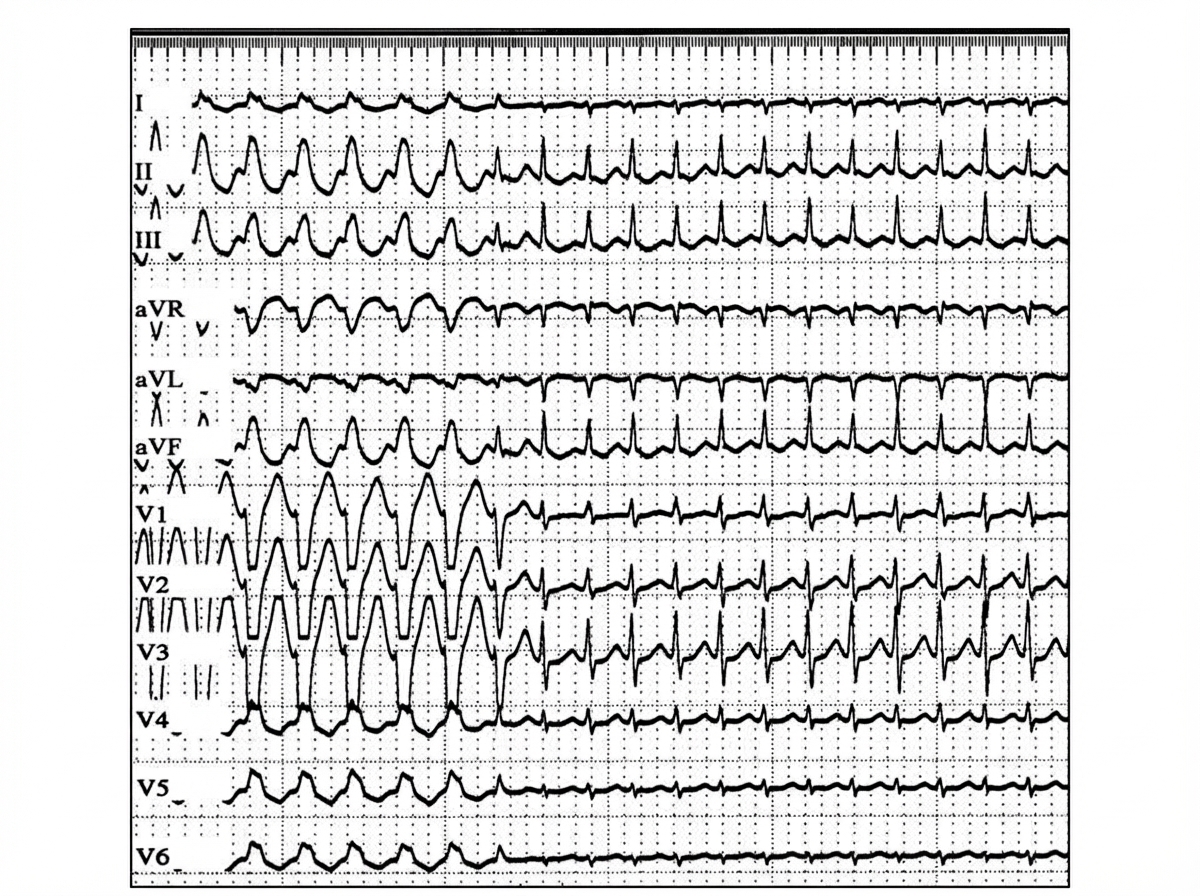
A 43-year-old woman develops acute renal failure following an emergency resection of a leaking abdominal aortic aneurysm. One week after surgery, the following laboratory values are obtained: Serum electrolytes (mEq/L): Na+ 127, K+ 5.9, Cl- 92, HCO3- 15. Blood urea nitrogen: 82 mg/dL. Serum creatinine: 6.7 mg/dL. The patient has gained 4 kg since surgery and is mildly dyspneic at rest. Eight hours after these values are reported, the following electrocardiogram is obtained. Which of the following is the most appropriate initial treatment in the management of this patient?
Which of the following is the primary acid-base disorder associated with hemorrhagic shock, diabetic ketoacidosis, and chronic renal failure?
What is the normal value of arterial HCO3' in gas exchange?
Practice by Chapter
Acid-Base Chemistry
Practice Questions
Respiratory Regulation of Acid-Base Balance
Practice Questions
Renal Regulation of Acid-Base Balance
Practice Questions
Bicarbonate Buffer System
Practice Questions
Non-Bicarbonate Buffer Systems
Practice Questions
Respiratory Acidosis and Alkalosis
Practice Questions
Metabolic Acidosis and Alkalosis
Practice Questions
Mixed Acid-Base Disorders
Practice Questions
Compensatory Mechanisms
Practice Questions
Clinical Assessment of Acid-Base Status
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
