Pharmacogenomics — MCQs

On this page
Which of the following is NOT an example of chronic adverse drug reactions?
The study of how variations in the human genome affect the response to medications is known as?
What is the approximate dose of a drug for a 3-year-old child compared to an adult dose?
In sub-acute toxicity studies, for what duration is the drug given to animals?
Administration of which of the following drugs requires therapeutic drug monitoring?
What are orphan drugs?
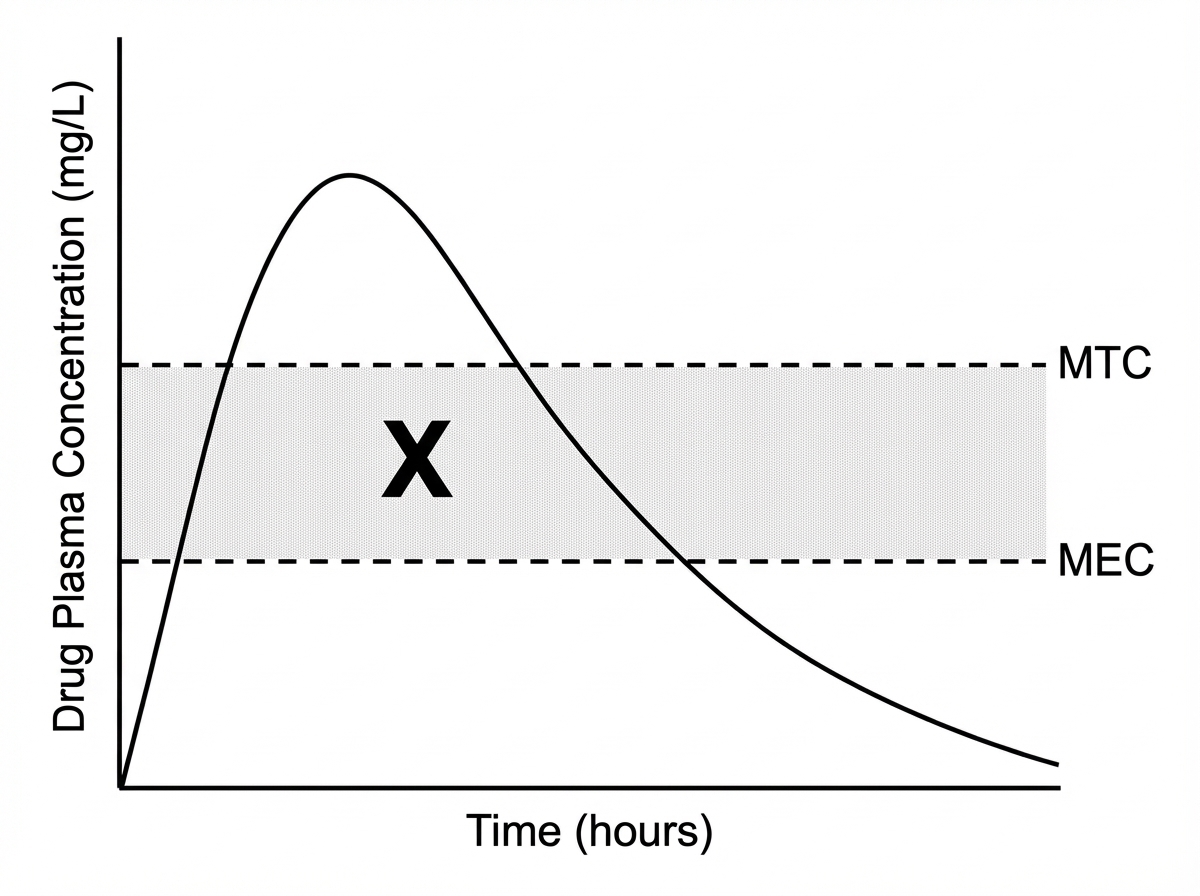
What does X represent in the given graph?
Which of the following drugs requires therapeutic drug monitoring?
A patient with history of ischemic stroke was started on clopidogrel. However, she had another attack of stroke after 6 months. Which of the following is likely to be responsible for the failure of clopidogrel in this patient?
Prolonged apnea may occur in patients with a genetically determined abnormal variant of cholinesterase following intravenous administration of
Practice by Chapter
Genetic Basis of Drug Response
Practice Questions
Pharmacogenomic Testing
Practice Questions
Cytochrome P450 Polymorphisms
Practice Questions
Pharmacogenomics of Drug Transporters
Practice Questions
Pharmacogenomics in Oncology
Practice Questions
Pharmacogenomics in Cardiovascular Therapeutics
Practice Questions
Pharmacogenomics in Psychiatry
Practice Questions
Personalized Medicine Approaches
Practice Questions
Ethical Considerations in Pharmacogenomics
Practice Questions
Implementation of Pharmacogenomics in Clinical Practice
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
