General Pharmacology — MCQs

On this page
What percentage of drugs exist as enantiomers?
What is the approximate number of patients recruited in phase II clinical trials of a drug?
Who synthesized the first H2 receptor blocker in 1972?
Ketanserin is:
What is characteristic of an agonist?
What is the typical outcome for a drug after undergoing Phase II biotransformation reactions?
Which of the following are NOT typically administered intradermally?
Which of the following routes of administration is shown in the given diagram?
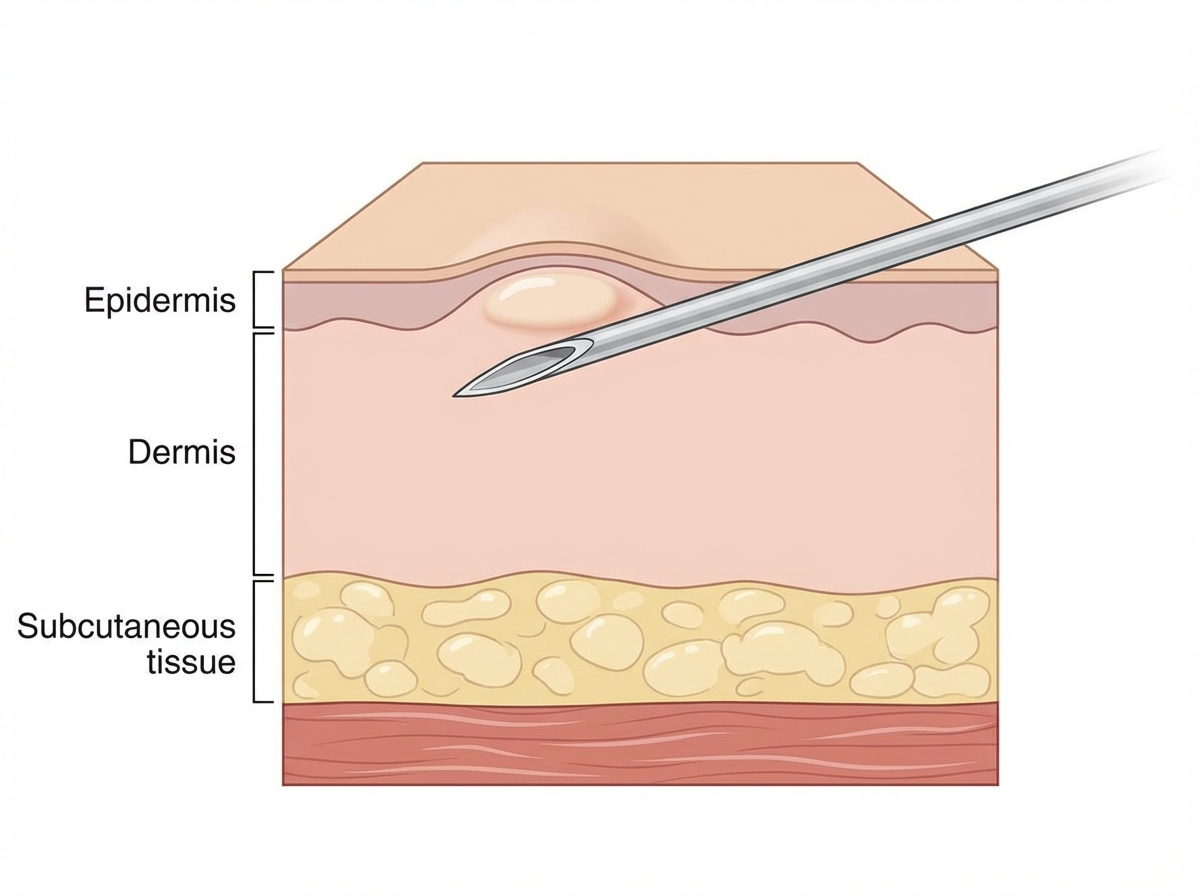
What is true about plasma half-life?
Which of the following is NOT a local route of drug administration?
Practice by Chapter
Pharmacokinetics: Absorption and Distribution
Practice Questions
Pharmacokinetics: Metabolism and Excretion
Practice Questions
Pharmacodynamics and Receptor Theory
Practice Questions
Drug-Receptor Interactions and Dose-Response
Practice Questions
Pharmacogenetics and Personalized Medicine
Practice Questions
Adverse Drug Reactions and Toxicity
Practice Questions
Drug Interactions
Practice Questions
Drug Development and Regulation
Practice Questions
Pediatric and Geriatric Pharmacology
Practice Questions
Placental Transfer and Lactation
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
