Drug Development and Regulation — MCQs

According to which of the following guidelines must a registered medical practitioner preserve medical records of patients for a minimum of 3 years from the last date of treatment?
A chemical is tested for carcinogenicity by examining its mutagenic effects on bacterial cells in culture. Which of the following tests is used to make this determination?
At which phase of a drug trial is permission from the DCGI required?
Which of the following combinations can result in severe toxicity due to inhibition of cytochrome P450 enzymes?
From which part of the Papaver somniferum plant does the latex, commonly referred to as 'milk', exude?
The following malformation in a baby due to drug intake by mother is classified as \qquad ADR?
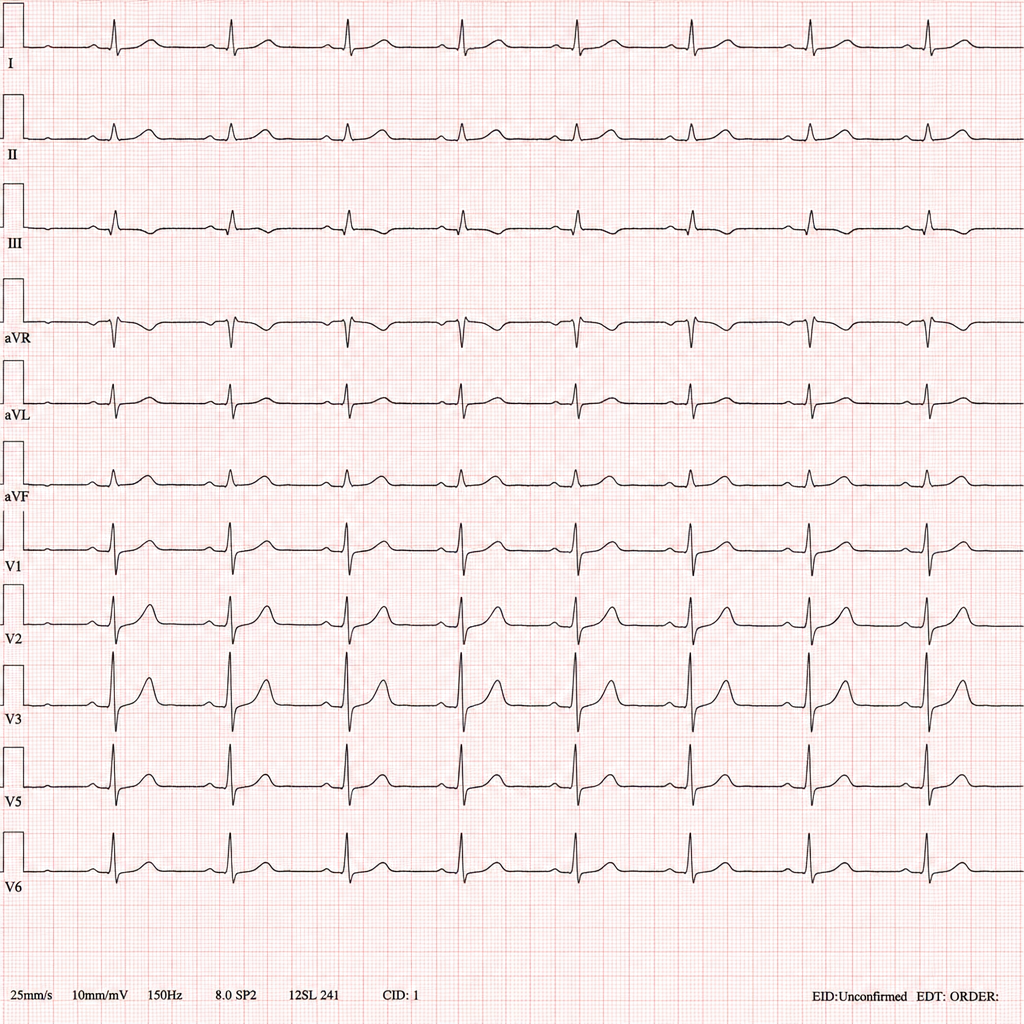
A 34-year-old male with opioid dependence enrolled in a maintenance programme presents to the emergency department with palpitations and two witnessed syncopal episodes over the past 24 hours. His only medication was started 6 weeks ago at a community addiction clinic. Baseline ECG on enrolment was normal. The on-call resident records the ECG shown in Image 1. Which drug is most likely responsible for the rhythm disturbance seen?
Which of the following drugs can be administered through all routes?
Therapeutic drug monitoring is done for all of the following drugs except?
Which of the following is an example of physiological antagonism?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
