Endocrine Pharmacology — MCQs

On this page
What is the drug of choice for a pregnant female suspected of having a baby with congenital adrenal hyperplasia?
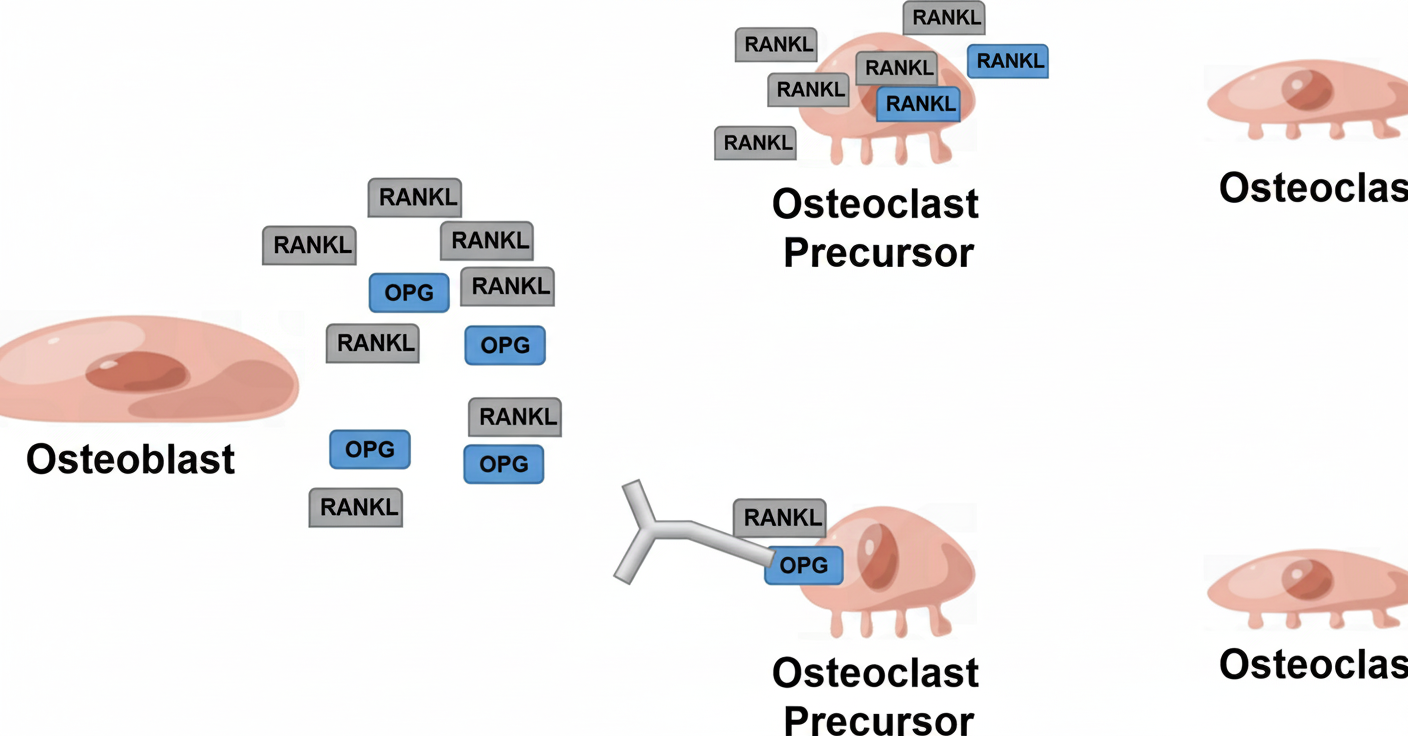
A drug used in the management of osteoporosis has the mechanism of action depicted in the accompanying figure. What is the likely identity of this drug?
What percentage of Lente insulin is amorphous?
Which of the following does not cause insulin release?
A 29-year-old male patient presents to the OPD with a 10-year history of coarse facial features and progressive enlargement of the hands and feet. Laboratory evaluation revealed elevated IGF-1 and non-suppressible growth hormone levels after the 75 g glucose challenge test. A diagnosis of acromegaly is made. Which of the following drugs is preferred for the management of this patient?
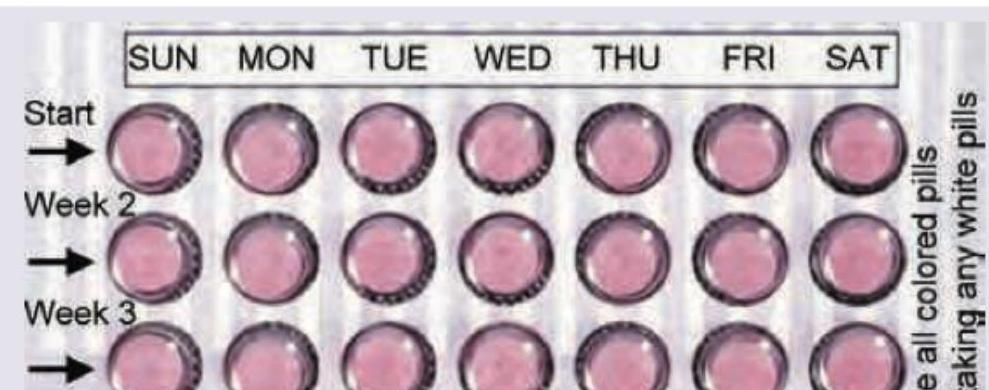
A 28-year-old woman is prescribed an oral contraceptive pill. The packaging shown in the image contains pills organized by weeks with different colored pills for Week 1, Week 2, and Week 3. What type of combined oral contraceptive preparation is this?
A 60-year-old post-menopausal woman presents with low back ache and pain in all legs muscles. Her lab reports show normal serum calcium, phosphate and normal serum alkaline phosphatase. For management of this condition, which of the following drugs is given by intranasal route?
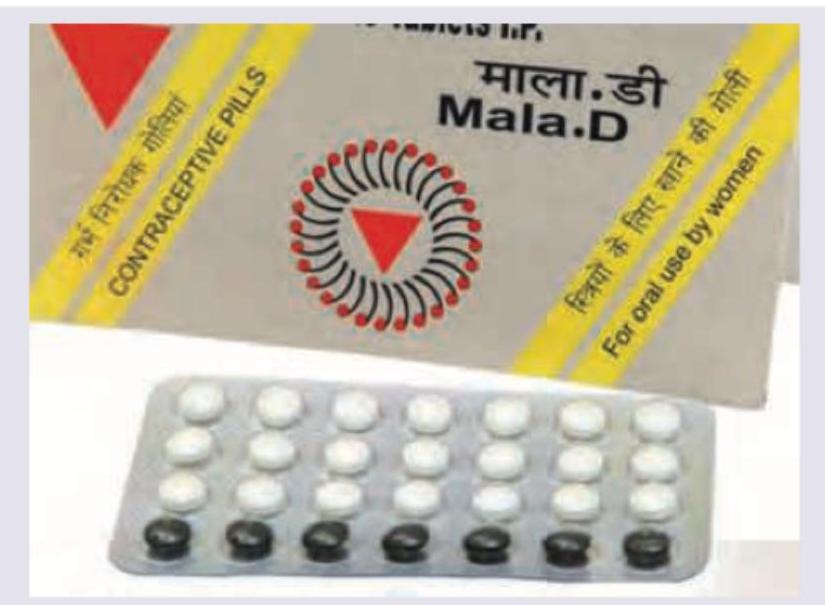
Select the correct composition of the OCP shown below:
Which of the following are major complications of oral pills? 1. Cholestasis jaundice 2. Chloasma and acne 3. Venous thromboembolism 4. Breast cancer Select the correct answer using the code given below.
All of the following are correct regarding drospirenone EXCEPT:
Practice by Chapter
Hypothalamic and Pituitary Hormones
Practice Questions
Thyroid Drugs and Antithyroid Agents
Practice Questions
Insulin and Oral Hypoglycemic Agents
Practice Questions
Adrenocorticosteroids
Practice Questions
Sex Hormones: Estrogens and Progestins
Practice Questions
Androgens and Anabolic Steroids
Practice Questions
Hormonal Contraceptives
Practice Questions
Drugs Affecting Calcium Metabolism
Practice Questions
Drugs for Osteoporosis
Practice Questions
Pharmacological Management of Obesity
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
