Drug Interactions — MCQs

On this page
Which of the following antiepileptic drugs does not cause oral contraceptive pill (OCP) failure?
Ciprofloxacin should not be given to an asthmatic using theophylline because:
Teeth stained by tetracycline appear _______ under UV light?
What is the basis for combining ritonavir with lopinavir?
Which of the following drugs does not increase the action of warfarin?
Which of the following drugs can lead to increased levels of theophylline?
A 54-year-old non-smoking male presents with a flulike illness. Initially treated symptomatically, he returns two days later still feeling unwell. His primary physician prescribes a macrolide along with symptomatic treatment. After five days, the patient reports persistent fever, increasing fatigue, weakness, and cough. He also experiences pain and limited motion in his right wrist, and left groin pain with difficulty ambulating. On physical exam, vital signs include pulse 110 bpm, temperature 102°F, respirations 24/min, and blood pressure 100/68 mm Hg. He appears weak and has lost 8 lb in 10 days. Lung examination reveals egophony and E to A changes in the left anterior and posterior fields. Physical findings include swelling with skin sloughing on the right wrist and tenderness with limited movement in the left groin. Laboratory data shows Hb 11 g/dL, Hct 33%, WBCs 16,000/uL with 90% neutrophils, BUN 42 mg/dL, creatinine 1.1 mg/dL, sodium 142 mEq/L, and potassium 3.4 mEq/L. Arterial blood gases on room air are: pH 7.45, PCO2 34 mm Hg, PO2 65 mm Hg. A chest X-ray is provided. Associated findings may include all of the following except?
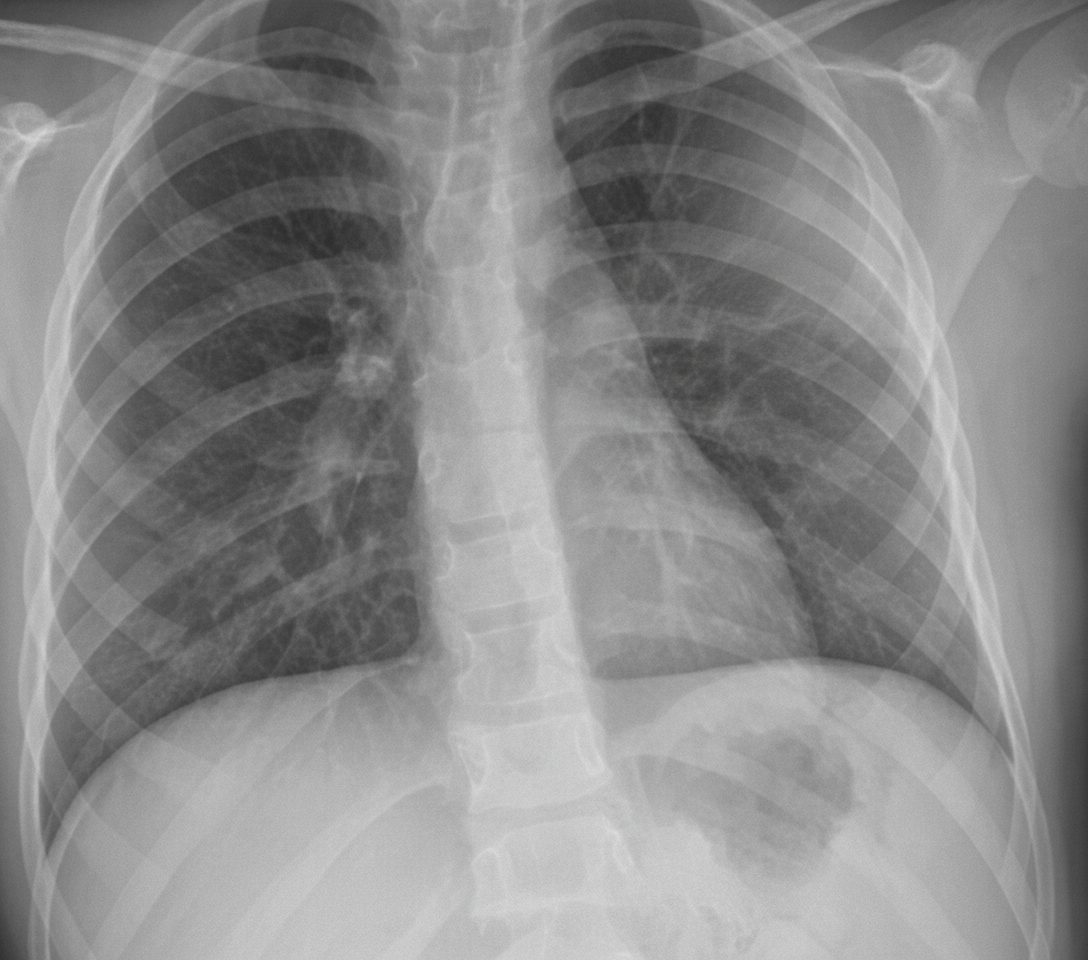
A 75-year-old male patient, a known case of pulmonary disease treated with inhalational corticosteroids and daily theophylline, was admitted for urinary retention. After catheterization and subsequent urinary tract infection treated with an antibiotic, the patient now presents with nausea, vomiting, abdominal pain, headache, fine hand tremor, and tachycardia. These symptoms are suspected to be due to increased serum levels of his medications. Which of the following drugs may be responsible for the patient's condition?
A hypertensive patient already receiving a drug 'X' to control his BP was prescribed a tricyclic antidepressant. This resulted in the abolition of the antihypertensive action of 'X'. Which of the following drug can be 'X'?
Which of the following medications is contraindicated in patients with allergy to sulphonamides?
Practice by Chapter
Mechanisms of Drug Interactions
Practice Questions
Pharmacokinetic Interactions
Practice Questions
Pharmacodynamic Interactions
Practice Questions
Drug-Food Interactions
Practice Questions
Drug-Disease Interactions
Practice Questions
Clinically Significant Drug Interactions
Practice Questions
Computer Systems for Detecting Drug Interactions
Practice Questions
Management of Drug Interactions
Practice Questions
Drug Interactions in Special Populations
Practice Questions
Role of P-glycoprotein in Drug Interactions
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
