Pharmacovigilance — MCQs

HIV sentinel surveillance is used for:
Which of the following statements about drug-induced SLE is NOT true?
A patient on warfarin has a high INR. Which drug likely caused this?
Therapeutic drug monitoring is done for:
Which of the following diseases is primarily monitored under the Integrated Disease Surveillance Program (IDSP)?
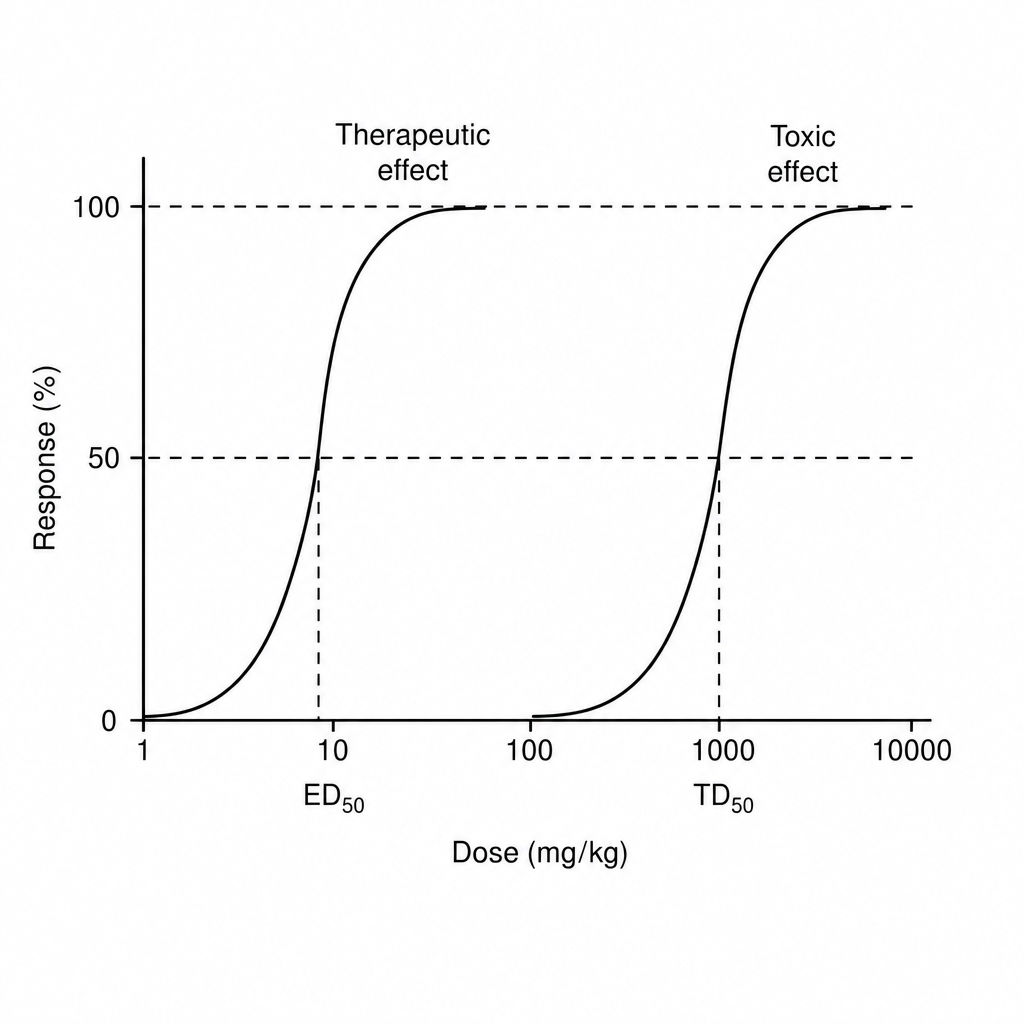
Find therapeutic index of drug from the information given below in the graph
According to Hill's criteria, which of the following is NOT a criterion for establishing causality in noncommunicable diseases?
A district shows API of 4.2, ABER 11%, and SPR 3.1%. What is the malaria surveillance status?
Cisapride was withdrawn from the market due to?
Permission from DCGI [Drug controller general, India] is needed before which phase of drug trial?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
