Clinical Pharmacology and Drug Toxicity — MCQs

On this page
Prescription of which of the following drugs can lead to periorbital edema of non-pitting variety?
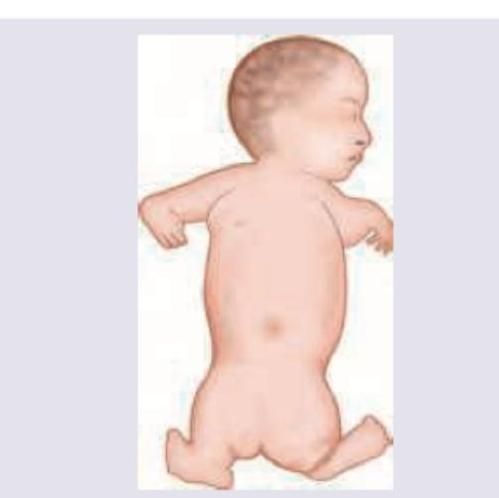
The following malformation in a baby due to drug intake by mother is classified as \qquad ADR?
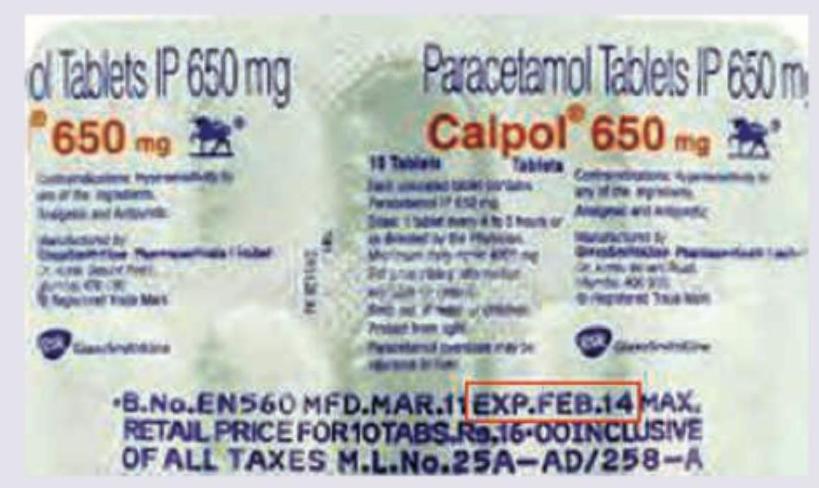
Which is correct about the marked area in the blister strip of Paracetamol (PCM)? (Recent NEET Pattern 2016-17)
A 65-year-old patient of dilated cardiomyopathy was prescribed 1 mg warfarin tablet for sustained atrial fibrillation. The chemist however gave him 2 mg tablets by mistake which resulted in him developing hemorrhagic complications (bleeding). CT scan is performed. This is which kind of ADR? (Recent NEET Pattern 2016-17) Warfarin Sodium Tablets, USP Crystalline 2 mg Protect from light
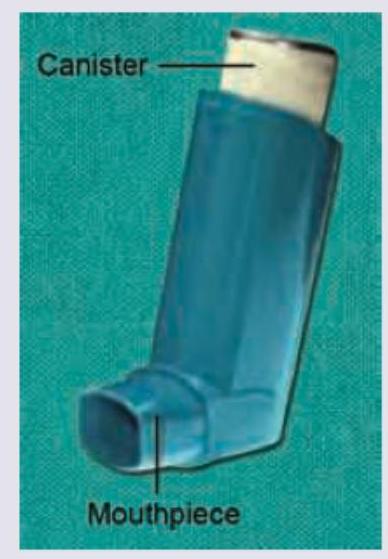
Which of the following instructions is not given if a second puff is required from this device?
A patient with rheumatic heart disease is in atrial fibrillation. The ECG shows irregularly irregular heart rhythm with absent P waves and down-sloping ST segment depression pronounced in V2-V3 and in the rhythm strip. The patient is experiencing general unwell being. Given the ECG findings and history, which medication is most likely causing the patient's symptoms?
A primigravida at 38 weeks pregnancy was put on oxytocin drip in view of slow labour at the rate of 30 mIU/min by the newly appointed registrar. She complains of confusion and starts throwing fits. What electrolyte imbalance is expected to have happened in this case?
The best drug for maintenance therapy of Systemic Lupus Erythematosus (SLE) during pregnancy is :
Detection of magnesium toxicity in a patient receiving magnesium sulphate is noticed by which of the following? I. Loss of tendon reflexes II. Increased respiratory rate III. Heart block, prolonged PR interval IV. Cardiac arrest Select the correct answer using the code given below :
Consider the following statements with regard to the treatment of vitamin A deficiency : I. Repeated high doses of retinol can cause liver damage and teratogenicity II. Acute overdose of vitamin A may lead to increased intracranial pressure and skin desquamation III. Regular vitamin A supplementation is also recommended for pregnant women even in countries where vitamin A deficiency is not endemic IV. Excessive intake of carotene may cause harmless orange pigmentation of the skin Which of the statements given above are correct?
Practice by Chapter
Principles of Clinical Pharmacology
Practice Questions
Therapeutic Drug Monitoring
Practice Questions
Drug Toxicity and Overdose
Practice Questions
Antidotes and Their Applications
Practice Questions
Management of Drug Poisoning
Practice Questions
Drug-Induced Liver Injury
Practice Questions
Drug-Induced Kidney Injury
Practice Questions
Drug-Induced Blood Dyscrasias
Practice Questions
Drug-Induced QT Prolongation
Practice Questions
Pharmacovigilance
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
