Clinical Pharmacology and Drug Toxicity — MCQs

On this page
Hemodialysis is not useful in which of the following conditions?
Which of the following congenital malformations is seen in a child born to a mother treated with oral anticoagulants?
Which of the following drugs can cause osteonecrosis when administered intravenously?
Sclerosing cholangitis is a known side effect caused by which of the following drugs?
What is the treatment for Type II paralysis in organophosphorus poisoning?
All of the following drugs require dose reduction in cirrhosis except?
What is the mechanism of action of pralidoxime?
Which of the following is used to treat cyanide poisoning?
What drug is associated with the following adverse drug reaction (ADR)?
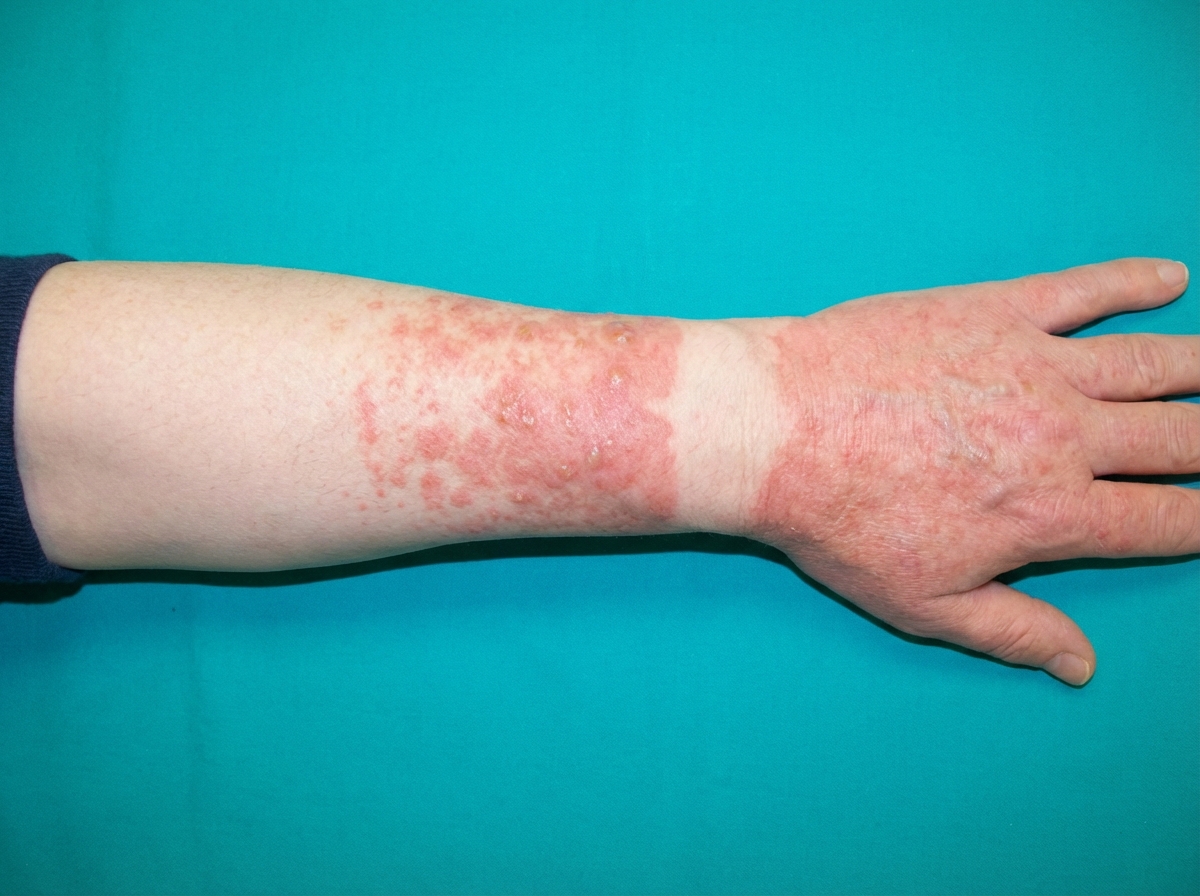
Which of the following drugs exhibits a therapeutic window phenomenon?
Practice by Chapter
Principles of Clinical Pharmacology
Practice Questions
Therapeutic Drug Monitoring
Practice Questions
Drug Toxicity and Overdose
Practice Questions
Antidotes and Their Applications
Practice Questions
Management of Drug Poisoning
Practice Questions
Drug-Induced Liver Injury
Practice Questions
Drug-Induced Kidney Injury
Practice Questions
Drug-Induced Blood Dyscrasias
Practice Questions
Drug-Induced QT Prolongation
Practice Questions
Pharmacovigilance
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
