Central Nervous System Pharmacology — MCQs

On this page
Erenumab was approved by FDA in 2018 for which condition?
All except one are centrally acting muscle relaxants. Which one is not?
Which of the following drugs has the highest potential to cause metabolic syndrome?
Which of the following medications are used for migraine prophylaxis?
Which of the following acts as an antagonist to the NMDA receptor?
Which of the following belongs to antifibrinolytic drugs?
Which of the following is an inverse agonist of the GABA receptor?
According to signal transduction mechanisms, what type of receptor is the GABA-B receptor?
A 30-year-old theatre actress developed facial wrinkles. The treating physician advised local injections of a drug indicated in cervical dystonia, spastic disorders like cerebral palsy, and prophylaxis of migraine. The physician warned of potential side effects including dry mouth and blurred vision. The actress identified the drug's site of action as depicted in the accompanying figure. Which of the following drugs is being described?
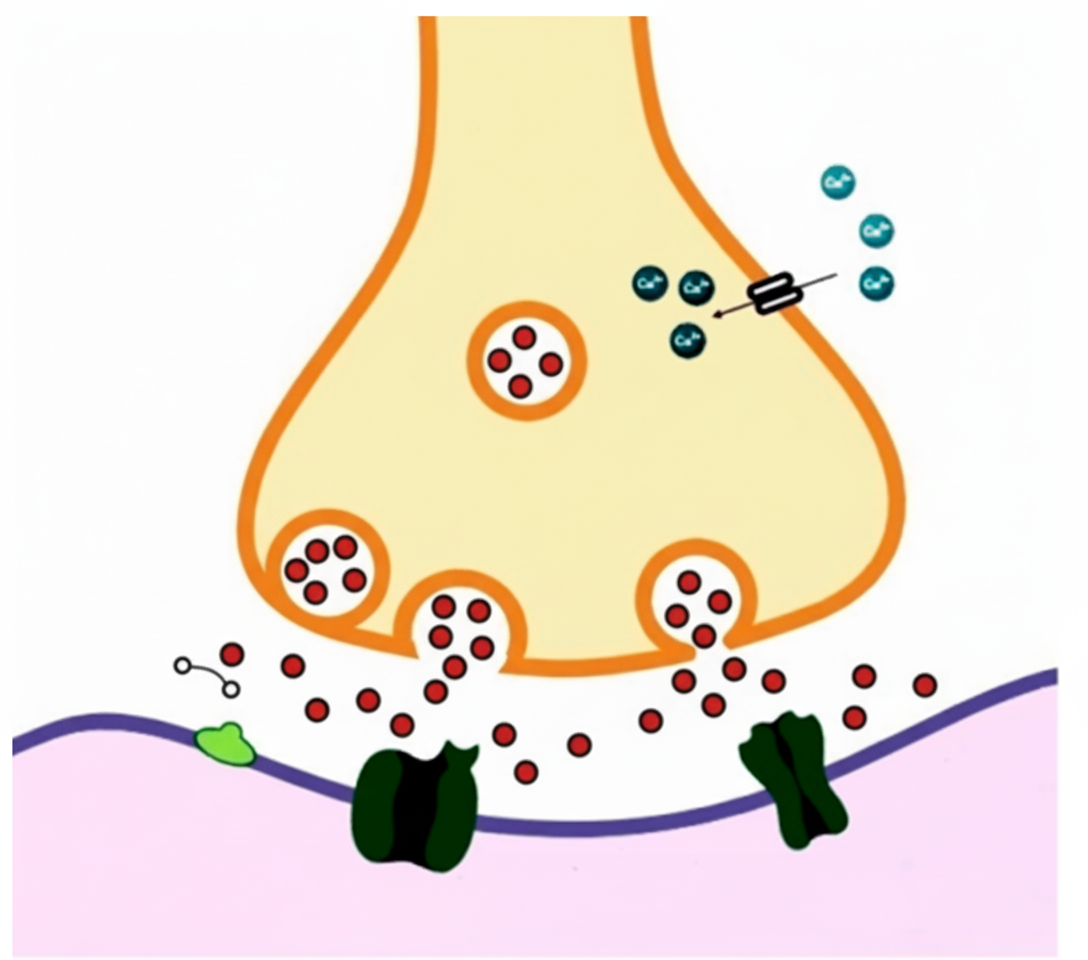
A 30-year-old male presents with a history of abnormal, excessive blinking and grunting sounds. He has no control over these symptoms, which have increased in frequency. Which of the following medications can be used for the treatment of this condition?
Practice by Chapter
General Anesthetics
Practice Questions
Local Anesthetics
Practice Questions
Sedative-Hypnotics
Practice Questions
Antiepileptic Drugs
Practice Questions
Antiparkinsonian Drugs
Practice Questions
Opioid Analgesics
Practice Questions
Drugs of Abuse and Addiction
Practice Questions
Psychostimulants
Practice Questions
Hallucinogens
Practice Questions
CNS Stimulants and Cognitive Enhancers
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
