Anticancer Drugs — MCQs

On this page
A patient on treatment for leukemia develops chest pain, pulmonary infiltrates, and pleural effusion. What is the likely cause?
Rituximab is an antibody against which of the following?
Gemcitabine is effective in which of the following conditions?
Temozolomide is used in the treatment of which of the following conditions?
Which of the following drugs produces significant nephrotoxicity?
A young male was diagnosed with acute myeloid leukemia and received induction chemotherapy with a doxorubicin-based regimen, which was successful. Two months later, he presents with swelling of both feet and dyspnea on exertion, along with paroxysmal nocturnal dyspnea. Which of the following is most likely responsible for this patient's symptoms?
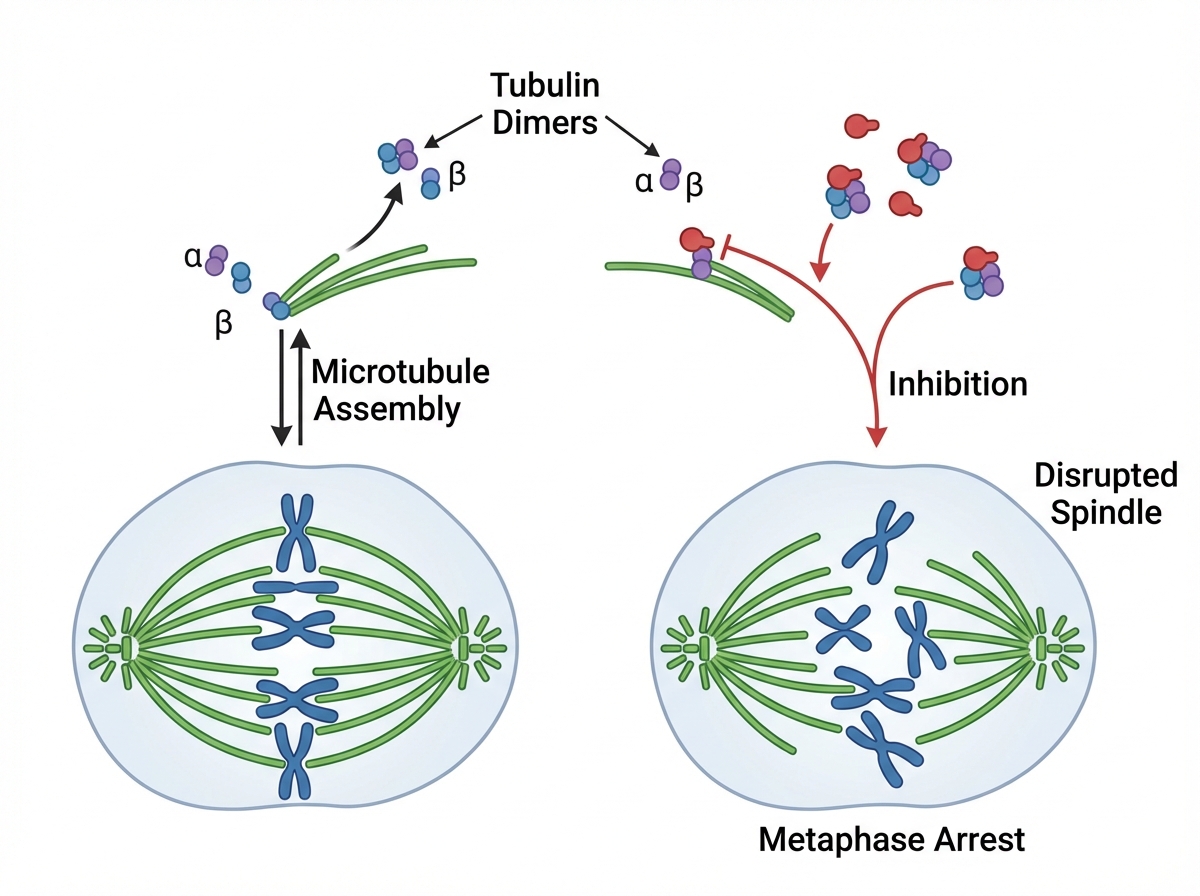
The provided figure illustrates the mechanism of action of which of the following drugs?
Cetuximab (an EGFR antagonist) can be used in which of the following conditions?
Which anticancer drug is known to cause nephrotoxicity?
Alkylating agents are known to cause which of the following conditions?
Practice by Chapter
Principles of Cancer Chemotherapy
Practice Questions
Alkylating Agents
Practice Questions
Antimetabolites
Practice Questions
Antitumor Antibiotics
Practice Questions
Plant Alkaloids
Practice Questions
Topoisomerase Inhibitors
Practice Questions
Hormonal Agents
Practice Questions
Targeted Therapy
Practice Questions
Immunotherapy
Practice Questions
Management of Chemotherapy Side Effects
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
