Nephrology — MCQs

On this page
What is the most common cause of urinary tract infection in children?
A 10-year-old boy presents with hypertension. There is no other significant history. What is the most likely cause of his hypertension?
A patient with congenital nephrotic syndrome requires which of the following procedures?
An 8-year-old child has had abdominal pain and dark urine for 10 days. Physical examination shows blotchy purple skin lesions on the trunk and extremities. Urinalysis shows hematuria and proteinuria. Serologic test results are negative for myeloperoxidase-antineutrophil cytoplasmic antibody (MPO-ANCA) and proteinase 3-antineutrophil cytoplasmic antibody (PR3-ANCA). A skin biopsy specimen shows necrotizing vasculitis of small dermal vessels. A renal biopsy specimen shows immune complex deposition in glomeruli, with some IgA-rich immune complexes. Which of the following is the most likely diagnosis?
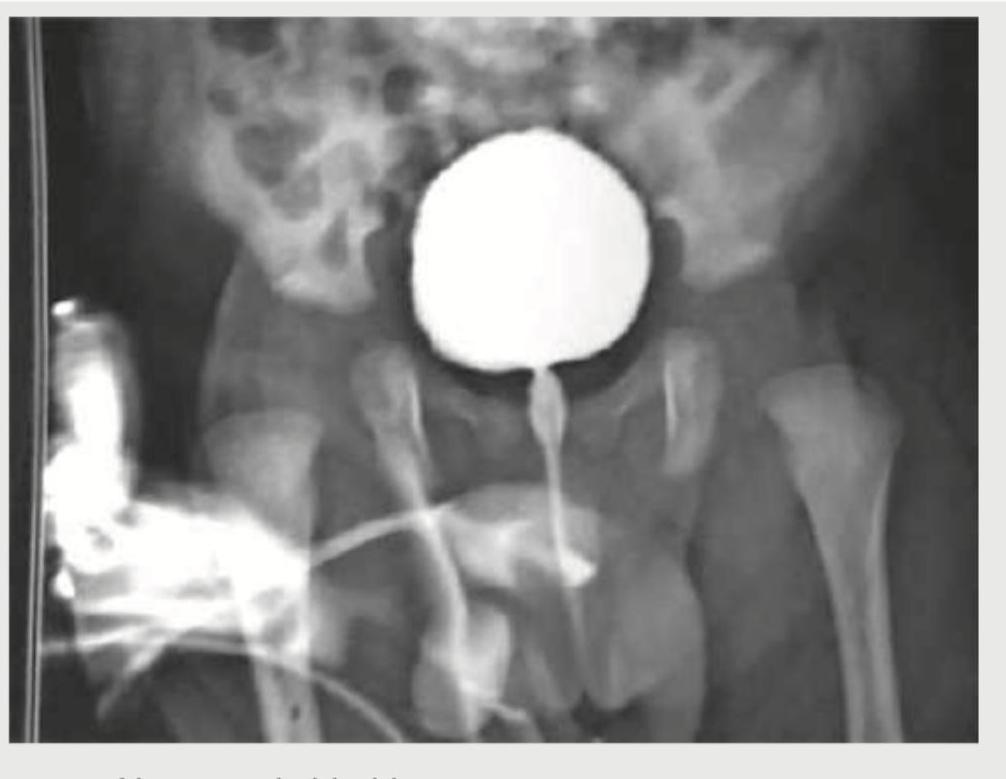
After her first urinary tract infection (UTI), a 1-year-old girl has a voiding cystourethrogram with findings shown below. Which of the following is the most appropriate management option?
Which of the following is NOT a characteristic of nephrotic syndrome in children?
A 9-year-old boy presents with decreased urine output, cola-colored urine, and swelling of the face and hands for 2 days. He is hypertensive, has a puffy face, and pitting edema of the lower limbs. He had a history of skin lesions 4 weeks prior. A diagnosis of post-streptococcal glomerulonephritis is made. At what point are C3 levels likely to return to normal?
A 3-year-old child presents with 3+ proteinuria, oliguria, and edema. There is no hematuria. What is the most likely diagnosis?
A 10-year-old girl presents with polyuria and polydipsia, along with hypokalemia, hypercalciuria, and metabolic alkalosis. What is the probable diagnosis?
A 2-year-old child presents with a 1-year history of generalized edema. His blood pressure is 107/70 mmHg. Urine examination shows hyaline casts and 3+ proteinuria, with no white blood cells or red blood cells. What is the likely diagnosis?
Practice by Chapter
Urinary Tract Infections
Practice Questions
Vesicoureteral Reflux
Practice Questions
Glomerulonephritis
Practice Questions
Nephrotic Syndrome
Practice Questions
Acute Kidney Injury
Practice Questions
Chronic Kidney Disease
Practice Questions
Renal Tubular Disorders
Practice Questions
Congenital Anomalies of the Kidney
Practice Questions
Hydronephrosis
Practice Questions
Hypertension in Children
Practice Questions
Hemolytic Uremic Syndrome
Practice Questions
Renal Replacement Therapy in Children
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
