Hematology — MCQs

On this page
Which of the following is most useful in the treatment of Kostmann's Syndrome?
Which chelating agent is used in chronic thalassemia major for iron overload?
A mother brings her 2-year-old boy with a history of pallor. Examination reveals pallor, petechiae, and splenomegaly. Which of the following is the most likely diagnosis?
Aplastic anemia with skeletal abnormalities, short stature, and pancreatic exocrine failure suggests which diagnosis?
Salmonellosis is most common in which of the following conditions?
Which of the following is characteristic of Fanconi Anemia?
In Sickle cell disease, aplastic crisis is due to which type of infection?
The peripheral blood smear of an anemic 1-year-old child is shown in the illustration. The most likely diagnosis is?
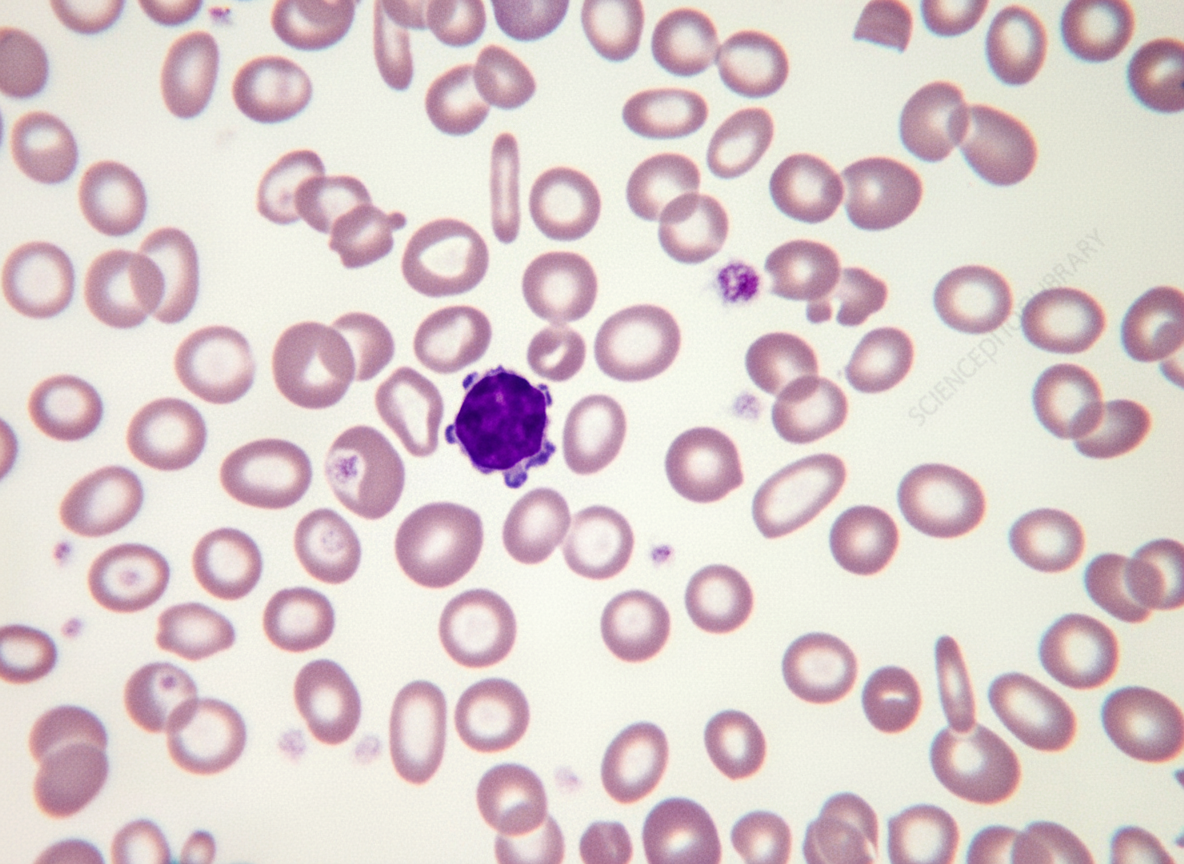
A 10-year-old boy presents with mucosal bleeding of 1 week's duration. Which investigation is most useful in this patient?
A newborn baby presented with profuse bleeding from the umbilical stump. What is the probable diagnosis?
Practice by Chapter
Anemias in Children
Practice Questions
Hemoglobinopathies
Practice Questions
Hemolytic Anemias
Practice Questions
Nutritional Anemias
Practice Questions
Thrombocytopenia
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
White Blood Cell Disorders
Practice Questions
Bone Marrow Failure Syndromes
Practice Questions
Blood Component Therapy
Practice Questions
Hemophilia and Von Willebrand Disease
Practice Questions
Evaluation of Bleeding Tendencies
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
