Hematology — MCQs

On this page
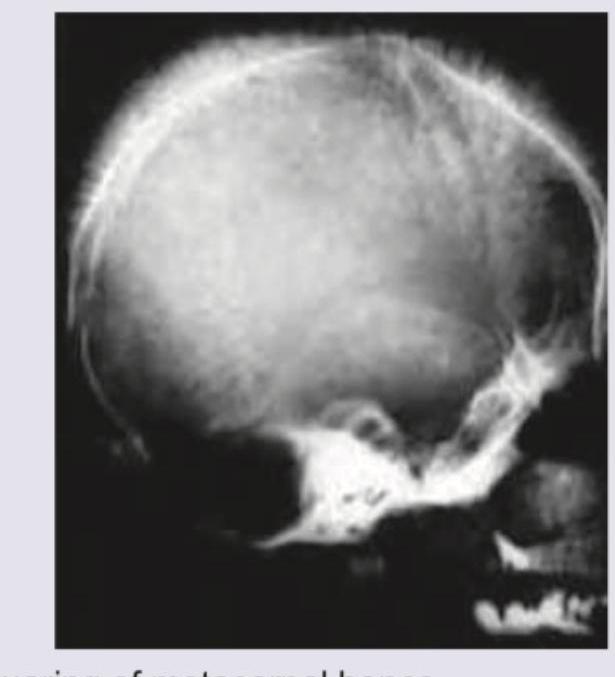
An 8-year-old boy with requirement of multiple blood transfusions. X-ray skull was performed. All are true about the condition shown except:
A 12-year-old child presents with acute kidney injury after a bout of dysentery. Not seen is:
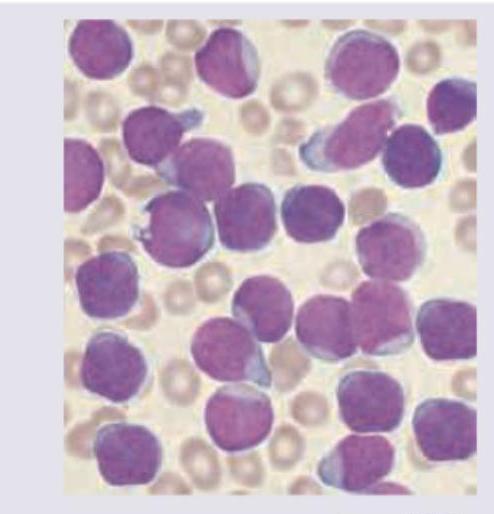
A 7-year-old boy presents with fever and weight loss. On examination he had pallor with lymphadenopathy. Peripheral smear is shown below. Diagnosis is: (AIIMS May 2017)
A 10-year-old boy presented with fatigue. Investigations revealed: Hemoglobin, 9 g/dL; MCV, 60 fL; MCH, 20 pg; and serum ferritin, 185 µg/L. The TLC was elevated and showed predominant lymphocytes and neutrophils. What is the likely diagnosis in this patient? **Normal values:** - Serum ferritin: 50-150 µg/L
Which of the following does not require a lumbar puncture in children?
A previously healthy child has sudden onset of red spots on body. There is a history of a preceding viral infection 1-4 weeks before the onset.
Normal reticulocyte count at birth is
Which of the following is the most common hematologic malignancy associated with Neurofibromatosis-1 (NF-1) in a child?
False about Shwachman-Diamond syndrome
Which of the following is most specific for congenital Rubella syndrome?
Practice by Chapter
Anemias in Children
Practice Questions
Hemoglobinopathies
Practice Questions
Hemolytic Anemias
Practice Questions
Nutritional Anemias
Practice Questions
Thrombocytopenia
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
White Blood Cell Disorders
Practice Questions
Bone Marrow Failure Syndromes
Practice Questions
Blood Component Therapy
Practice Questions
Hemophilia and Von Willebrand Disease
Practice Questions
Evaluation of Bleeding Tendencies
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
