Hematology — MCQs

On this page
An 18-month-old boy presented with a tender, swollen, warm right knee with significant hemarthrosis after an injury. His PT is 12 seconds (normal, 13 seconds), PTT is over 100 seconds (normal, 25 seconds), platelet count is 300,000/mm 3, and bleeding time is normal. What should be the initial management?
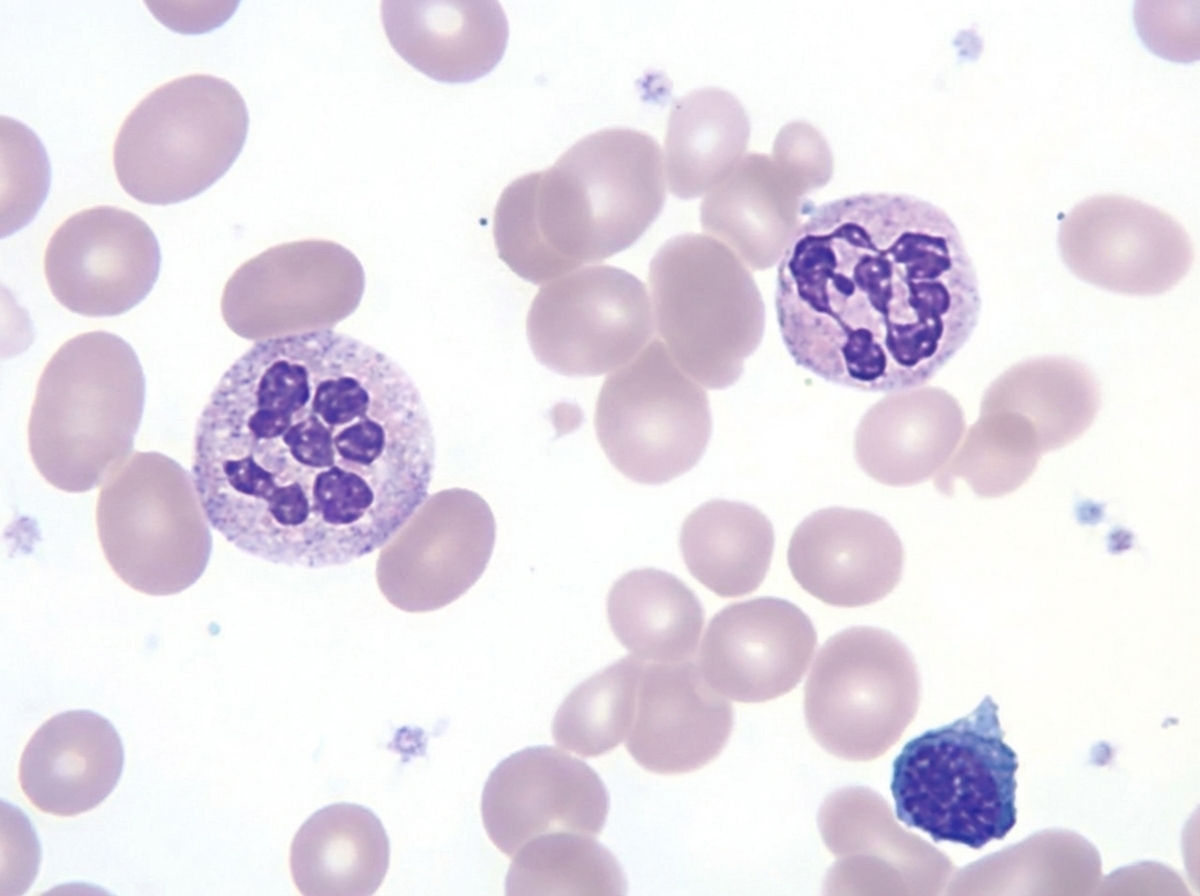
An otherwise healthy 1-year-old child has a routine CBC revealing polymorphonuclear neutrophils as shown below. Which of the following is the most appropriate next step?
A 5-year-old child presents with a history of intermittent fever for the past 2 weeks, petechial spots all over the body, and increasing pallor for the past 1 month. Examination reveals splenomegaly 2 cm below the costal margin. What is the most likely diagnosis?
What is true about Immune Thrombocytopenic purpura (ITP)?
A child presents with Hb of 6.5 gm%, MCV of 65, MCH of 15, and protoporphyria with a much reduced red cell distribution width. What is the most likely diagnosis?
Autosplenectomy is seen in which of the following conditions?
Diamond Blackfan anemia is another name for which of the following conditions?
In a newborn with ABO incompatibility, what is the characteristic feature observed on the peripheral smear?
Which finding is abnormal in the classic presentation of hemorrhagic disease of the newborn?
A 15-year-old boy presented with a one-day history of bleeding gums, subconjunctival bleed, and purpuric rash. Investigations revealed the following results: Hb - 6.4 gm/dL; TLC - 26,500/mm3; Platelets - 35,000/mm3; Prothrombin time - 20 sec (control 13 sec); Partial thromboplastin time - 50 sec; and Fibrinogen - 10 mg/dL. Peripheral smear was suggestive of acute myeloblastic leukemia. Which of the following is the most likely diagnosis?
Practice by Chapter
Anemias in Children
Practice Questions
Hemoglobinopathies
Practice Questions
Hemolytic Anemias
Practice Questions
Nutritional Anemias
Practice Questions
Thrombocytopenia
Practice Questions
Bleeding Disorders
Practice Questions
Thrombotic Disorders
Practice Questions
White Blood Cell Disorders
Practice Questions
Bone Marrow Failure Syndromes
Practice Questions
Blood Component Therapy
Practice Questions
Hemophilia and Von Willebrand Disease
Practice Questions
Evaluation of Bleeding Tendencies
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
