Vascular Pathology — MCQs

On this page
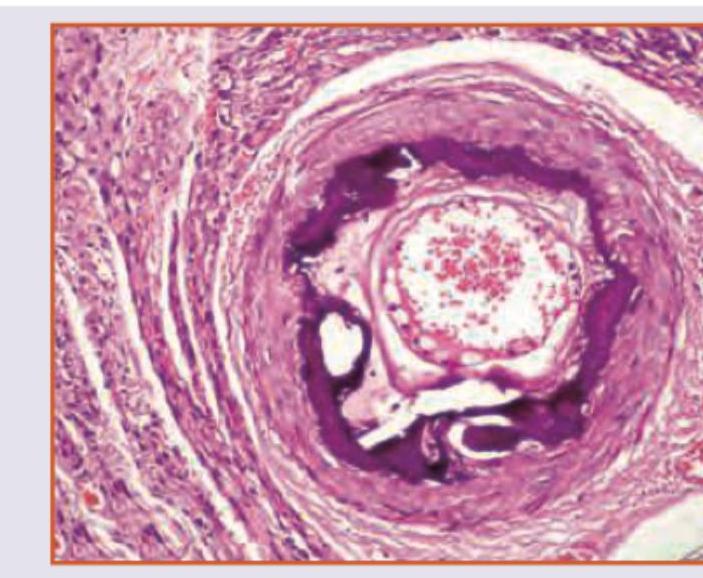
Which is true about the image shown?
Which of the following are the malignancies associated with lymphoedema? I. Kaposi Sarcoma II. Squamous cell carcinoma III. Malignant melanoma IV. Leukaemia Select the correct answer using the code given below :
Which of the following are correct about glomus tumour? 1. It arises from Suquet-Hoyer canals. 2. Its usual site is nail bed. 3. It is usually a small purple nodule. 4. It is painless.
The malignancy associated with Stewart-Treves syndrome is
A 50-year-old man presents with headache, chest discomfort, and blurred vision. His headache started 2 days ago and has not improved. He describes it as severe, throbbing, localized to the occipital part of the head and worse at the end of the day. He says he has associated nausea but denies any vomiting. Past medical history is significant for hypertension diagnosed 15 years ago, managed with beta-blockers until the patient self d/c’ed them a month ago. He has not seen a physician for the past 2 years. Family history is significant for hypertension and an ST-elevation myocardial infarction in his father and diabetes mellitus in his mother. Vitals signs are a blood pressure of 200/110 mm Hg, a pulse rate of 100/min and respiratory rate of 18/min Ophthalmoscopy reveals arteriolar nicking and papilledema. His ECG is normal. Laboratory findings are significant for a serum creatinine of 1.4 mg/dL and a blood urea nitrogen of 25 mg/dL. Urinalysis has 2+ protein. He is started on intravenous nitroprusside. Which of the following best explains the pathophysiology responsible for the neovascular changes present in this patient?
Berry aneurysm most commonly occurs due to?
Which is associated with vasculitis of medium sized vessels
In polyarteritis nodosa, aneurysms are seen in all organs EXCEPT:
True about Henoch-Schonlein purpura are the following, EXCEPT:
Which one of the following does not cause small vessel vasculitis?
Practice by Chapter
Atherosclerosis
Practice Questions
Hypertensive Vascular Disease
Practice Questions
Aneurysms and Dissection
Practice Questions
Vasculitis
Practice Questions
Venous Disease and Thrombosis
Practice Questions
Vascular Tumors
Practice Questions
Varicose Veins and Lymphatics
Practice Questions
Pathology of Vascular Interventions
Practice Questions
Vascular Diseases in Specific Organs
Practice Questions
Congenital Vascular Anomalies
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
