Pleural Diseases — MCQs

Best marker for distinguishing reactive from neoplastic mesothelial proliferation?
Most common organism causing SBP after E. coli is
Which of the following conditions is least likely to cause pneumothorax?
All the following in the Light's criteria are suggestive of exudative pleural effusion except.
Decreased glucose level in pleural effusion is found in:
Which of the following conditions can produce hemothorax?
Mesothelioma is closely associated with which of the following?
What occurs in the case of pneumothorax?
Ferruginous bodies are seen in:
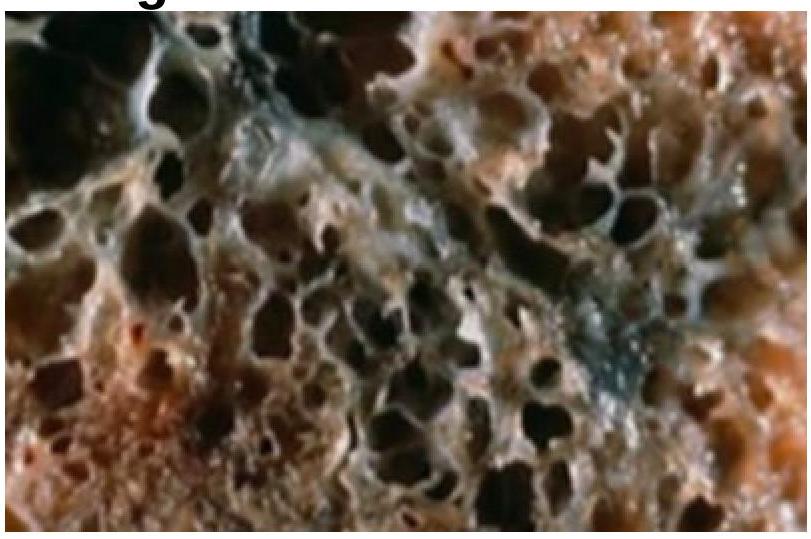
A 35-year-old woman with a long history of dyspnea, chronic cough, sputum production, and wheezing dies of respiratory failure following a bout of lobar pneumonia. She was not a smoker or an alcoholic. Which of the following underlying conditions is most likely associated with the pathologic changes shown in the lung autopsy?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
