Respiratory Pathology — MCQs

On this page
Asbestosis of the lung is associated with all of the following except?
Which of the following is the most characteristic feature of Adult Respiratory Distress Syndrome (ARDS)?
What is the characteristic histopathological finding in shock lung?
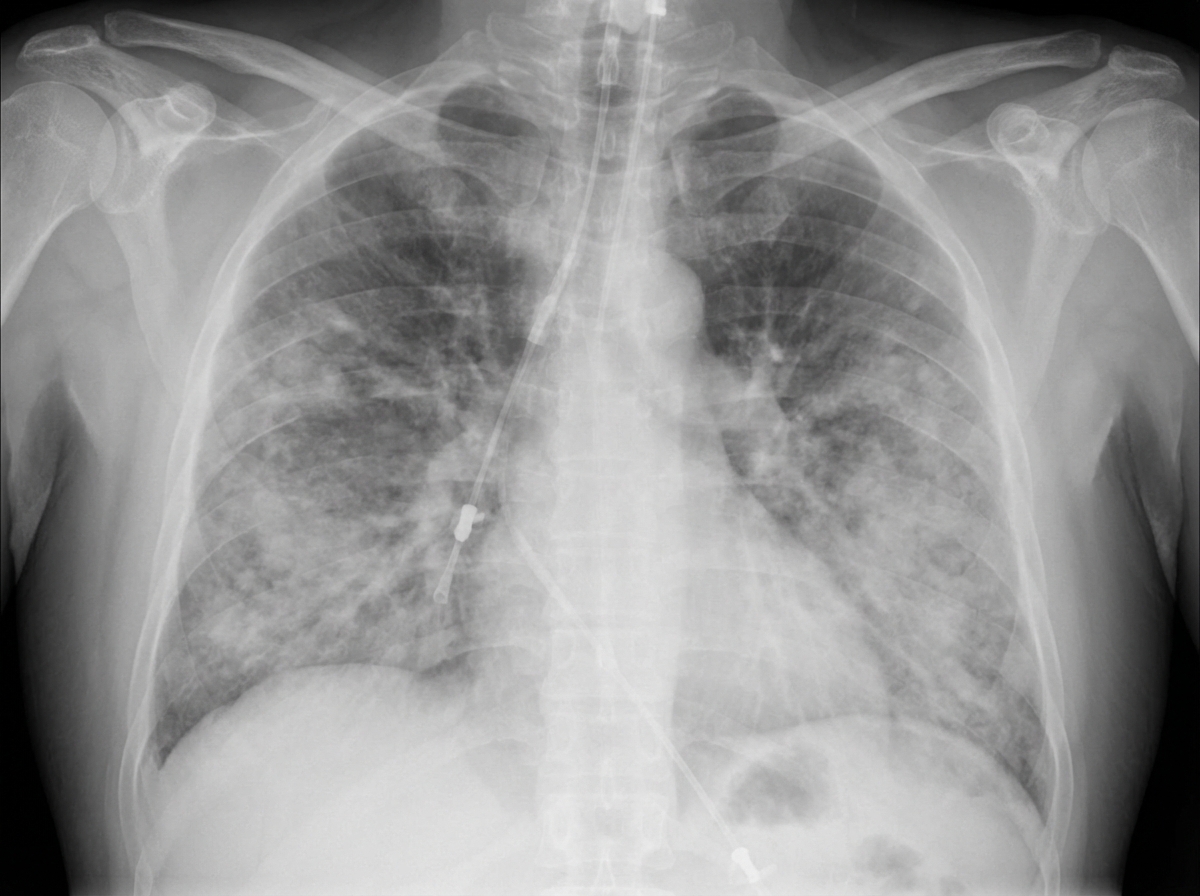
What is the pathophysiological cause of the disease shown in the X-ray and gross specimen from an ICU patient?
The term "brown induration" is used in relation to which of the following organs?
In the course of a laryngoscopic examination for hoarseness, a small lesion is found on the true vocal cord of a 57-year-old male smoker. On biopsy, severe squamous dysplasia is noted. If untreated, this lesion may progress to which of the following?
BAL of a patient shows foamy macrophages with decreased CD4:CD8 ratio. What is the diagnosis?
Curschmann's spirals in sputum are seen in which of the following conditions?
What is the most common presentation associated with thymoma?
A 50-year-old man dies of a chronic respiratory illness characterized by dyspnea, cough, and wheezing expiration over many years. His symptoms, initially episodic, increased in frequency and became continuous and intractable at the time of death. Which of the following is the most likely histologic finding in the lungs at autopsy?
Practice by Chapter
Congenital Anomalies
Practice Questions
Atelectasis and Acute Lung Injury
Practice Questions
Obstructive Pulmonary Diseases
Practice Questions
Restrictive Pulmonary Diseases
Practice Questions
Lung Infections
Practice Questions
Pulmonary Vascular Diseases
Practice Questions
Lung Tumors
Practice Questions
Pleural Diseases
Practice Questions
Interstitial Lung Diseases
Practice Questions
Occupational Lung Diseases
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
