Respiratory Pathology — MCQs

On this page
A 56-year-old chronic smoker has a mass in the bronchus resected. What is the possible marker for this type of tumor?
What is a feature of shock lung?
Pleural fibroma is differentiated from mesothelioma by the presence of which of the following markers in the former?
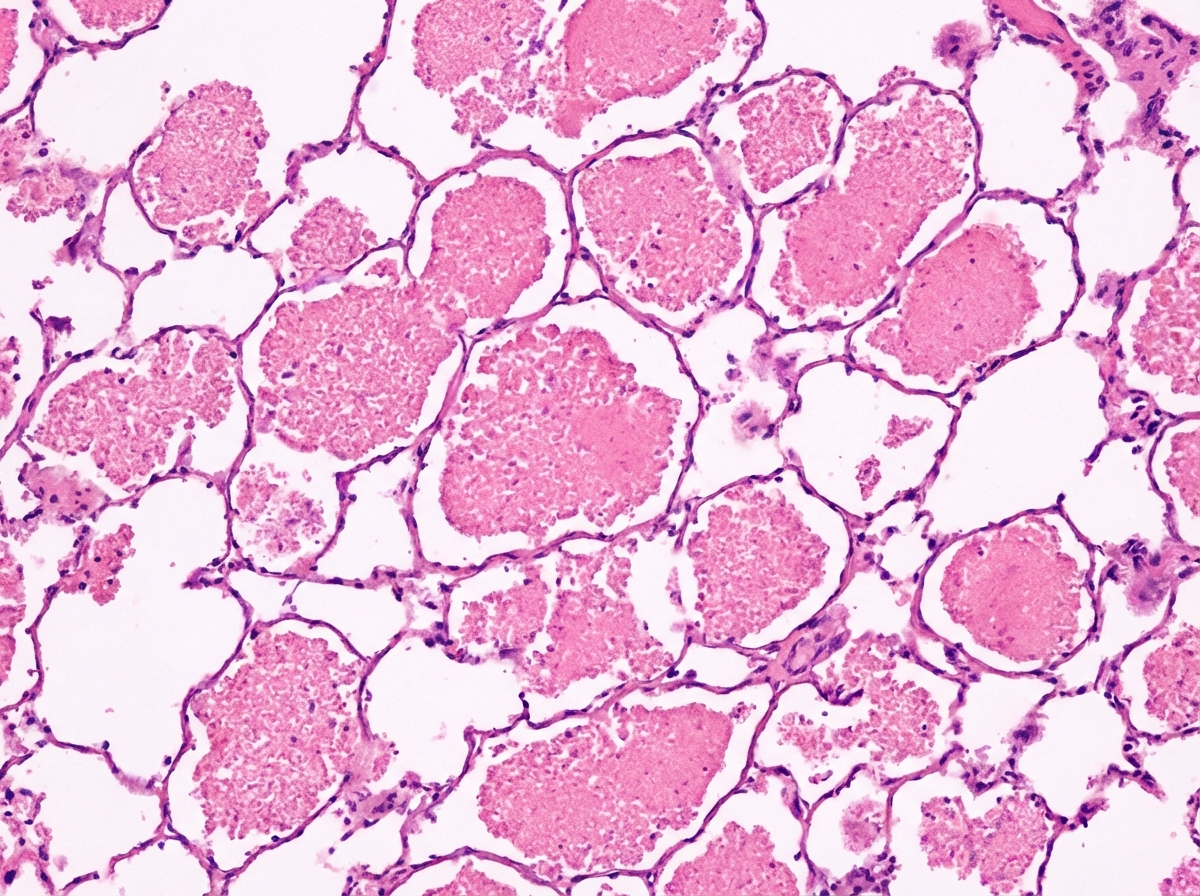
A full-term neonate develops severe progressive respiratory distress shortly after birth and subsequently dies. The image shows post-mortem lung biopsy findings. What is the most likely diagnosis?
True about panacinar emphysema?
Autopsy of a 23-year-old male victim of a motor vehicle accident reveals a small cluster of caseating granulomas in the right lung just above the interlobar fissure and similar granulomas in the hilar lymph nodes. Acid-fast staining demonstrates acid-fast bacilli within these lesions. No other lesions were found in the remaining organs and systems. Which of the following is the MOST accurate interpretation of these findings?
"Honeycomb lung" is most typical of which condition?
All of the following statements regarding centriacinar emphysema are true, except:
Pleural mesothelioma is associated with which of the following?
What is the most common clinical type of emphysema?
Practice by Chapter
Congenital Anomalies
Practice Questions
Atelectasis and Acute Lung Injury
Practice Questions
Obstructive Pulmonary Diseases
Practice Questions
Restrictive Pulmonary Diseases
Practice Questions
Lung Infections
Practice Questions
Pulmonary Vascular Diseases
Practice Questions
Lung Tumors
Practice Questions
Pleural Diseases
Practice Questions
Interstitial Lung Diseases
Practice Questions
Occupational Lung Diseases
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
