Respiratory Pathology — MCQs

On this page
Which of the following is known to cause cavitating lung lesions?
Distension of distant alveoli is seen in which type of emphysema?
All of the following statements are true regarding idiopathic pulmonary fibrosis, except:
In which of the following forms of pulmonary carcinoma is the classic progression of metaplasia to dysplasia to carcinoma in situ observed?
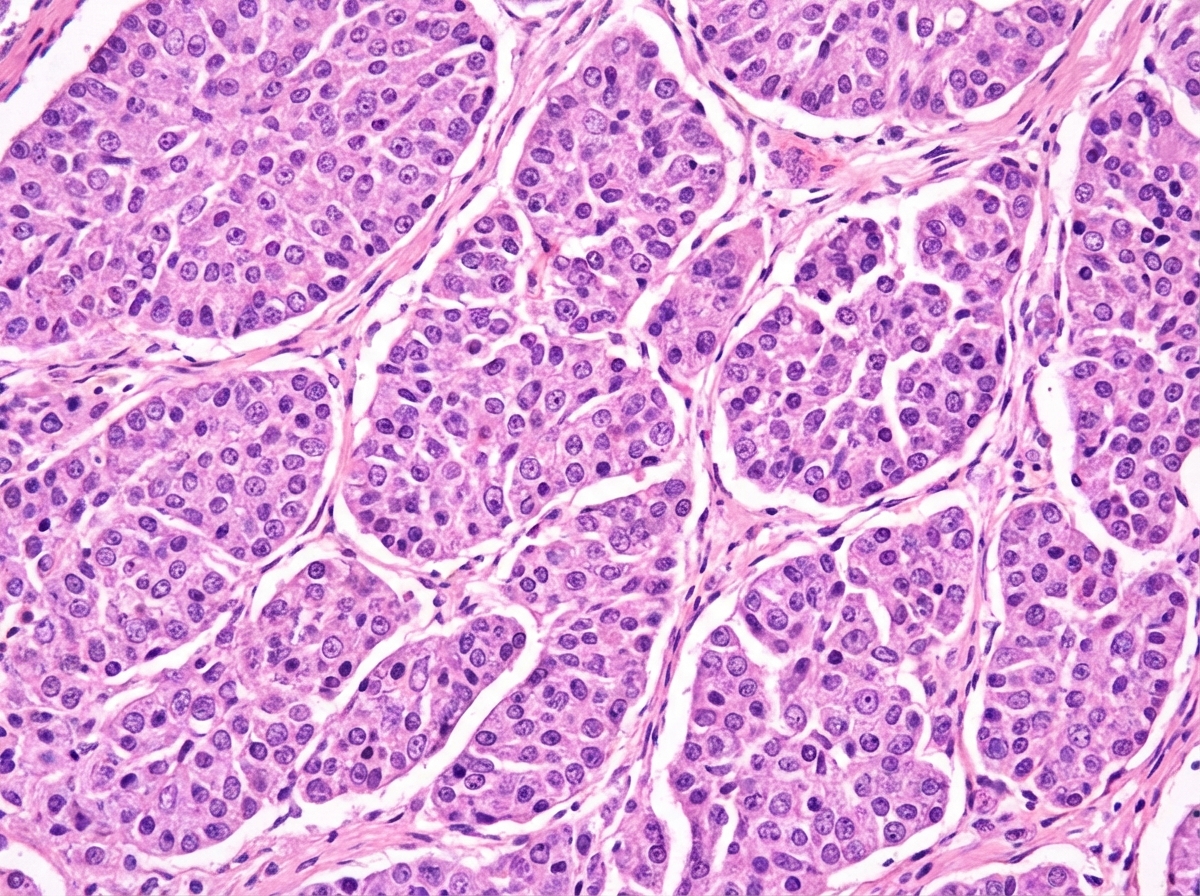
A 30-year-old woman presents with a history of fever for 2 weeks and multiple episodes of haemoptysis. Bronchoscopy shows a polypoid mass covered with intact mucosa. Surgical resection was performed, and the histology slide is shown below. What is the diagnosis?
Goodpasture's syndrome is characterized by?
A 60-year-old male with a long-standing history of breathlessness and heavy smoking since the age of 20 presents with an emaciated, anxious appearance and pursed-lip breathing, but without cyanosis. The chest is barrel-shaped. The primary pathology of the lung in this patient is associated with which of the following pathogenetic mechanisms?
Cavitation of the lungs is not a feature of which of the following conditions?
Histologic sections of lung tissue from an individual with adult respiratory distress syndrome (ARDS) are most likely to reveal what finding?
A study of HIV-infected persons shows that those with CD4+ lymphocyte counts below 100 cells/mL are at increased risk for pulmonary infections. Some patients present with concurrent hepatosplenomegaly, lymphadenopathy, malabsorption with weight loss, night sweats, and fever. Bronchoalveolar lavage specimens examined microscopically reveal macrophages filled with acid-fast infectious organisms. Which of the following infections have these persons developed?
Practice by Chapter
Congenital Anomalies
Practice Questions
Atelectasis and Acute Lung Injury
Practice Questions
Obstructive Pulmonary Diseases
Practice Questions
Restrictive Pulmonary Diseases
Practice Questions
Lung Infections
Practice Questions
Pulmonary Vascular Diseases
Practice Questions
Lung Tumors
Practice Questions
Pleural Diseases
Practice Questions
Interstitial Lung Diseases
Practice Questions
Occupational Lung Diseases
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
