Respiratory Pathology — MCQs

On this page
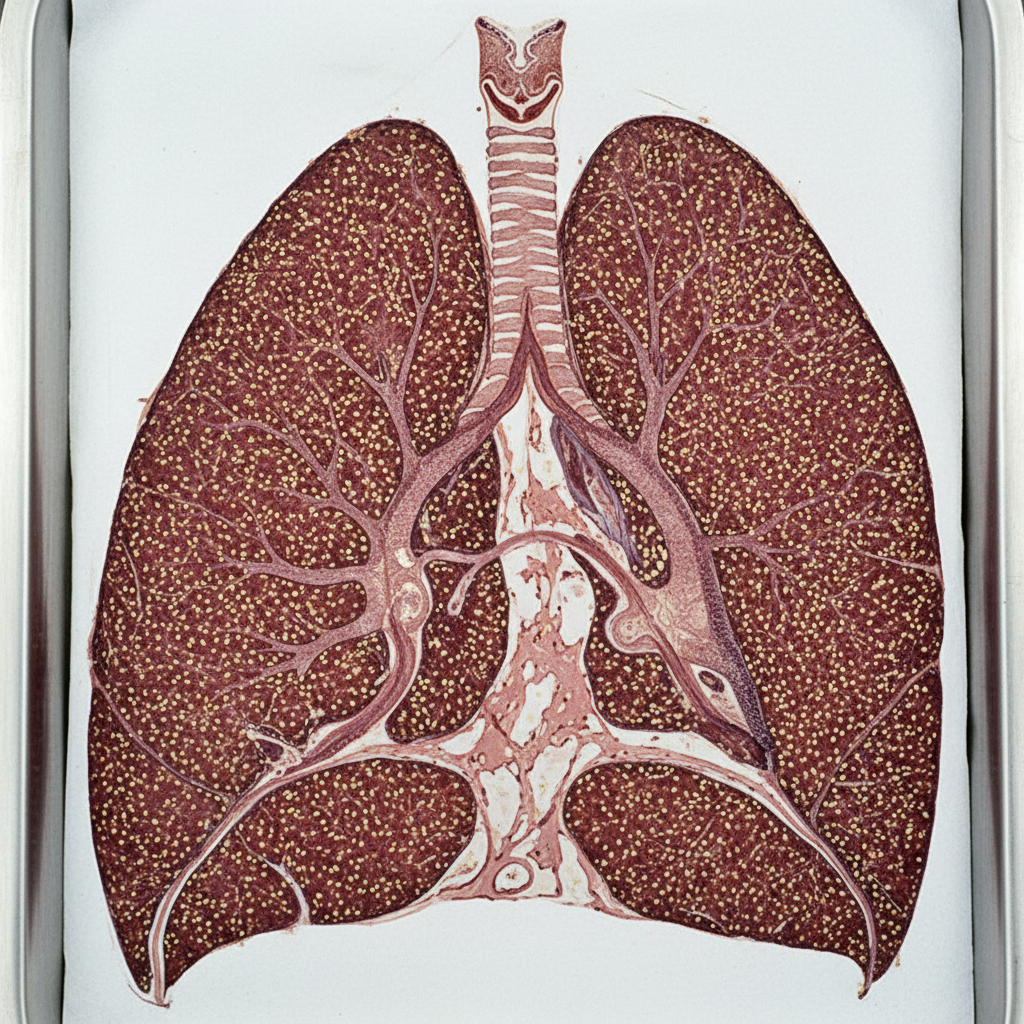
A patient underwent lung transplantation. Below is the gross appearance of the post-mortem lung. What is the most likely diagnosis?
What is the causative particle in asbestosis?
Infraclavicular lesion of tuberculosis is known as?
Which condition is characterized by alveolar hemorrhage and hemosiderin-laden macrophages?
Alpha-1-antitrypsin deficiency is associated with which type of emphysema?
Which of the following is a finding in biopsy of mesothelioma of pleura?
The alveoli are filled with exudate and the air is displaced, converting the lung into a solid organ. This description suggests:
Lymphangitis carcinomatosa is a typical feature of which of the following?
What is the synonym for shock lung?
Clubbing is the least common in which of the following conditions?
Practice by Chapter
Congenital Anomalies
Practice Questions
Atelectasis and Acute Lung Injury
Practice Questions
Obstructive Pulmonary Diseases
Practice Questions
Restrictive Pulmonary Diseases
Practice Questions
Lung Infections
Practice Questions
Pulmonary Vascular Diseases
Practice Questions
Lung Tumors
Practice Questions
Pleural Diseases
Practice Questions
Interstitial Lung Diseases
Practice Questions
Occupational Lung Diseases
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
