Respiratory Pathology — MCQs

On this page
What statement is FALSE regarding the Reid index?
All of the following statements are true regarding primary ciliary dyskinesia, except:
A 56-year old male, with a 20-year history of working in an asbestos factory, presented with a lesion at the lung apex. Electron microscopy of a lung biopsy revealed characteristic findings. What is your diagnosis?
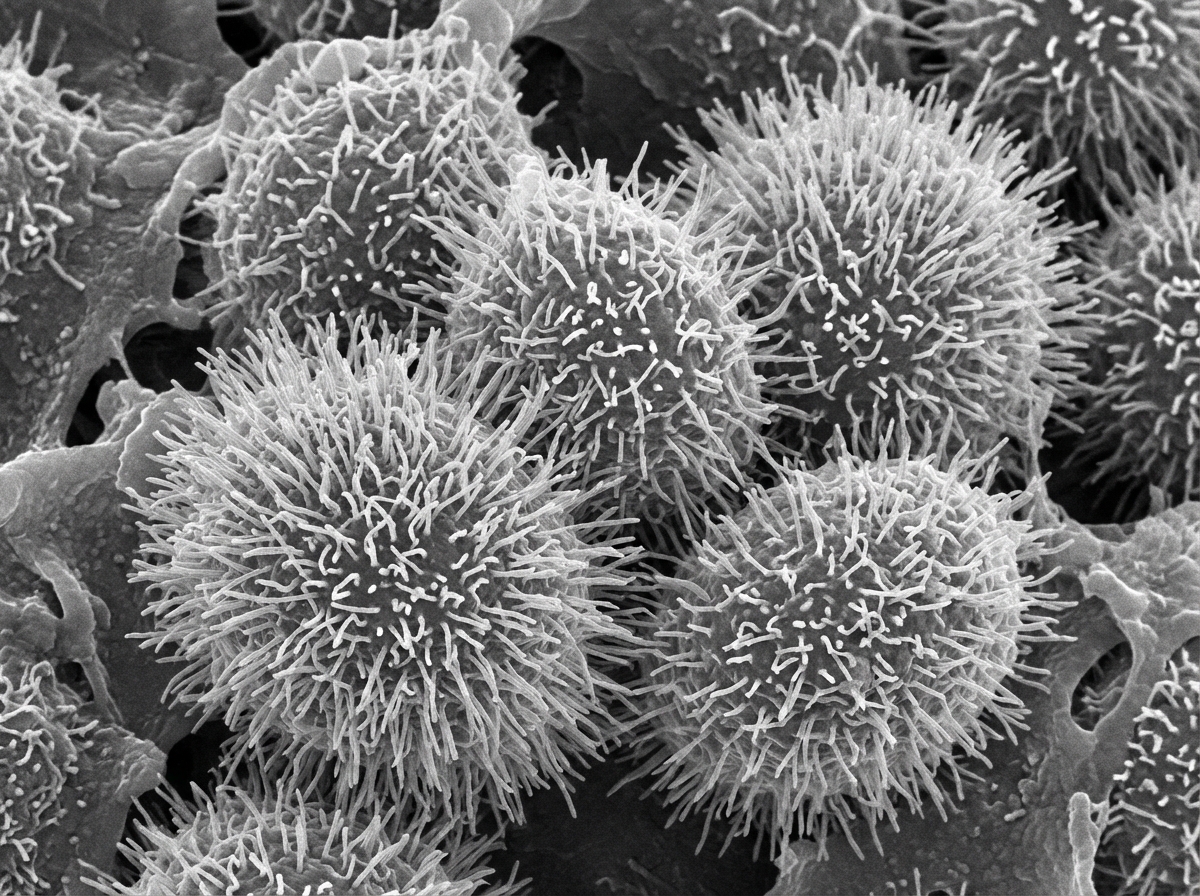
Sputum from an asthma patient may show which of the following findings?
Radon-222 is believed to be a risk factor for which of the following cancers?
The Azzopardi effect is a characteristic finding associated with which of the following lung tumors?
Which type of lung carcinoma is most commonly associated with producing superior vena cava syndrome?
Small cell carcinoma commonly metastasizes to which of the following organs?
Sputum analysis of an asthma patient shows Charcot Leyden crystals. From where are these crystals derived from?
Which of the following is true about silicosis?
Practice by Chapter
Congenital Anomalies
Practice Questions
Atelectasis and Acute Lung Injury
Practice Questions
Obstructive Pulmonary Diseases
Practice Questions
Restrictive Pulmonary Diseases
Practice Questions
Lung Infections
Practice Questions
Pulmonary Vascular Diseases
Practice Questions
Lung Tumors
Practice Questions
Pleural Diseases
Practice Questions
Interstitial Lung Diseases
Practice Questions
Occupational Lung Diseases
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
